The author's 90G reef aquarium.

Photo is from the Reef2Reef archives and courtesy of Greg Gdowski, ©2019, All Rights Reserved.
We put a lot of chemicals into our tanks. Many manufacturers tell you to base additions of these chemicals on your tank's volume – but what is that anyway?
What is your tank volume?
In the beginning, this won’t matter that much. Most of us just add up the stated volume of all of our components. In my case, I have a 90-gallon tank, a 60-gallon refugium, and a 30-gallon sump. That’s 180 gallons. Done, why go further?
As you go forward, every product you will put in your tank will give you a dosage (e.g. 1 drop per 100 l of water daily). When I started down the path of creating a Balling method tank, the issue of “tank volume” was always used extremely loosely. Did they mean: 1) system volume, 2) display tank volume, or 3) actual water volume. It confused the heck out of me until I stepped back and realized that every time I put something into the water, it was all about the eventual concentration of that chemical in the water.
It’s sort of like making your favorite cocktail (why does every article I write refer to alcohol?). If you put in twice as much vodka, you will notice it. For some, that is good, for others, perhaps not. The same is true for your corals. It’s really just best to get the recipe correct.
So, what is your water volume? Seems easy, right? You have sand, corals, rocks, filters, skimmers, pvc pipes, heaters, among other things occupying the space in your stated tank volume that are indeed NOT water. How much volume is that? How much are you off? More importantly, how in the world are you going to figure that out?
If you are like me, you were so excited to fill the tank on the first day that you didn’t even think of measuring how much water you put in. Now you are in a conundrum; it's full of water with fish and corals. You are certainly not going to empty it to figure that out. What are you going to do? That is what this short article is all about.
Once you hear the solution, you will likely say, why didn’t I think of that? For me, it was more like, why didn’t I think of that 10 years ago. I’m sure I’m not the first one to think of this solution, but I thought it was worthwhile to put it out there, likely, again.
Periodic Table of Elements
This image is a royalty-free image from Pixabay.
Using chemical tests as a method for determining water volume.
Here it is in a nutshell. I put in a specific amount of chemical into my tank. I measure the chemical concentration, before and after I put the chemical into the tank. I use the resulting change in concentration to estimate the water volume based on the manufacturers stated dosing prescription.
Let’s run through an example.
I occasionally dose calcium chloride in my tank to set the overall Ca+ level in my tank. I use Red Sea Reef Foundation A (CoralVue). Their stated dosing prescription is as follows:
1 gram will raise the Ca level of 100 liters (26.4 gallons) by 3.6ppm.
Translating that dosage into a formula yields;
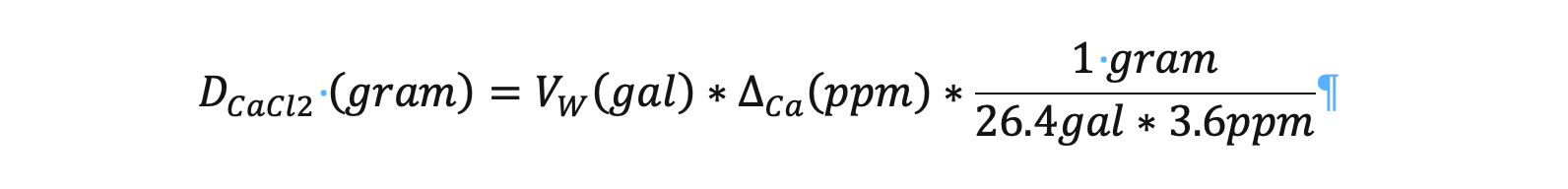
Where: VW is the volume of water, DCa is the change in Ca+ concentration, and DCaCl2 is the dosage of calcium chloride.
Let’s say you measured the Ca+ level in your tank and it was 370ppm. Being on the low side, you might want to raise the Ca+ level in your tank by 50ppm. Based on the formula, you might be inclined to dose 94.7 grams in my 180-gallon system in order to raise the concentration by 50ppm. If the estimate of your tank water volume was too high, you might end up raising the concentration by more than 50ppm.
The best way to do this is by using a two-dose strategy that results in first estimating your water volume. Start with dosing an amount that is substantially lower than you think you would need, which you can accurately measure using your scale. I started with 60 grams. I then measured the change in concentration as the difference between the Calcium tests that I did before (370ppm) I added the dose to the tank and after (405ppm) I added the dose to the tank.
Let’s rearrange the formula and solve for water volume:

Based on the outcome of the first dose, I still need to dose additional calcium chloride in order to raise the calcium by another 15ppm in order to achieve 420ppm. This is done by using the new estimate of water volume (163 gallons).

There are a lot of formulas on this page, but it’s really not all that complicated. In the end, had I put in 94.7 grams of calcium chloride, I would have only been off by less than five grams. That would have raised the Ca concentration by 55.2ppm instead of 50ppm. I ended up putting in 60+25.7 grams because this little experiment showed that I actually have a total volume of 163 gallons instead of my initial estimate of 180 gallons.
As you read this, you are probably saying that was a lot of effort to eliminate about 10% of error. I concur that you would be correct, in the short term. However, when you start dosing over long periods of time, small errors perpetuate and grow over time. After you do this once, everything you dose will be closer to what the manufacturer intended for you to put in the tank!
Until next time,
Happy reefing!
Greg
References
CoralVue. "Red Sea Reef Foundation A. Red Sea’s CALCIUM+ contains blended salts of calcium and strontium in the ratio required by corals and is part of Red Sea’s complete Reef Care Program.". from https://www.coralvue.com/red-sea-reef-foundation-a-1kg-powder.
~~~~~~~~~~~
We encourage all our readers to join the Reef2Reef forum. It’s easy to register, free, and reefkeeping is much easier and more fun in a community of fellow aquarists. We pride ourselves on a warm and family-friendly forum where everyone is welcome. You will also find lots of contests and giveaways with our sponsors.
~~~~~~~~~~~~
Author Profile: Greg Gdowski, Ph.D.
Greg has 20 years of aquarium experience, and he has been keeping reef aquariums for the past 10 years. A photo of his current 90G is at the top of this article. He and his wife are also both dog lovers and have two special-needs Vizslas at home.
Greg is also the Executive Director of the Center for Medical Technology and Innovation and Associate Professor in the Department of Biomedical Engineering at University of Rochester.
Photo is from the Reef2Reef archives and courtesy of Greg Gdowski, ©2019, All Rights Reserved.
We put a lot of chemicals into our tanks. Many manufacturers tell you to base additions of these chemicals on your tank's volume – but what is that anyway?
What is your tank volume?
In the beginning, this won’t matter that much. Most of us just add up the stated volume of all of our components. In my case, I have a 90-gallon tank, a 60-gallon refugium, and a 30-gallon sump. That’s 180 gallons. Done, why go further?
As you go forward, every product you will put in your tank will give you a dosage (e.g. 1 drop per 100 l of water daily). When I started down the path of creating a Balling method tank, the issue of “tank volume” was always used extremely loosely. Did they mean: 1) system volume, 2) display tank volume, or 3) actual water volume. It confused the heck out of me until I stepped back and realized that every time I put something into the water, it was all about the eventual concentration of that chemical in the water.
It’s sort of like making your favorite cocktail (why does every article I write refer to alcohol?). If you put in twice as much vodka, you will notice it. For some, that is good, for others, perhaps not. The same is true for your corals. It’s really just best to get the recipe correct.
So, what is your water volume? Seems easy, right? You have sand, corals, rocks, filters, skimmers, pvc pipes, heaters, among other things occupying the space in your stated tank volume that are indeed NOT water. How much volume is that? How much are you off? More importantly, how in the world are you going to figure that out?
If you are like me, you were so excited to fill the tank on the first day that you didn’t even think of measuring how much water you put in. Now you are in a conundrum; it's full of water with fish and corals. You are certainly not going to empty it to figure that out. What are you going to do? That is what this short article is all about.
Once you hear the solution, you will likely say, why didn’t I think of that? For me, it was more like, why didn’t I think of that 10 years ago. I’m sure I’m not the first one to think of this solution, but I thought it was worthwhile to put it out there, likely, again.
Periodic Table of Elements
This image is a royalty-free image from Pixabay.
Using chemical tests as a method for determining water volume.
Here it is in a nutshell. I put in a specific amount of chemical into my tank. I measure the chemical concentration, before and after I put the chemical into the tank. I use the resulting change in concentration to estimate the water volume based on the manufacturers stated dosing prescription.
Let’s run through an example.
I occasionally dose calcium chloride in my tank to set the overall Ca+ level in my tank. I use Red Sea Reef Foundation A (CoralVue). Their stated dosing prescription is as follows:
1 gram will raise the Ca level of 100 liters (26.4 gallons) by 3.6ppm.
Translating that dosage into a formula yields;
Where: VW is the volume of water, DCa is the change in Ca+ concentration, and DCaCl2 is the dosage of calcium chloride.
Let’s say you measured the Ca+ level in your tank and it was 370ppm. Being on the low side, you might want to raise the Ca+ level in your tank by 50ppm. Based on the formula, you might be inclined to dose 94.7 grams in my 180-gallon system in order to raise the concentration by 50ppm. If the estimate of your tank water volume was too high, you might end up raising the concentration by more than 50ppm.
The best way to do this is by using a two-dose strategy that results in first estimating your water volume. Start with dosing an amount that is substantially lower than you think you would need, which you can accurately measure using your scale. I started with 60 grams. I then measured the change in concentration as the difference between the Calcium tests that I did before (370ppm) I added the dose to the tank and after (405ppm) I added the dose to the tank.
Let’s rearrange the formula and solve for water volume:
Based on the outcome of the first dose, I still need to dose additional calcium chloride in order to raise the calcium by another 15ppm in order to achieve 420ppm. This is done by using the new estimate of water volume (163 gallons).
There are a lot of formulas on this page, but it’s really not all that complicated. In the end, had I put in 94.7 grams of calcium chloride, I would have only been off by less than five grams. That would have raised the Ca concentration by 55.2ppm instead of 50ppm. I ended up putting in 60+25.7 grams because this little experiment showed that I actually have a total volume of 163 gallons instead of my initial estimate of 180 gallons.
As you read this, you are probably saying that was a lot of effort to eliminate about 10% of error. I concur that you would be correct, in the short term. However, when you start dosing over long periods of time, small errors perpetuate and grow over time. After you do this once, everything you dose will be closer to what the manufacturer intended for you to put in the tank!
Until next time,
Happy reefing!
Greg
References
CoralVue. "Red Sea Reef Foundation A. Red Sea’s CALCIUM+ contains blended salts of calcium and strontium in the ratio required by corals and is part of Red Sea’s complete Reef Care Program.". from https://www.coralvue.com/red-sea-reef-foundation-a-1kg-powder.
~~~~~~~~~~~
We encourage all our readers to join the Reef2Reef forum. It’s easy to register, free, and reefkeeping is much easier and more fun in a community of fellow aquarists. We pride ourselves on a warm and family-friendly forum where everyone is welcome. You will also find lots of contests and giveaways with our sponsors.
~~~~~~~~~~~~
Author Profile: Greg Gdowski, Ph.D.
Greg has 20 years of aquarium experience, and he has been keeping reef aquariums for the past 10 years. A photo of his current 90G is at the top of this article. He and his wife are also both dog lovers and have two special-needs Vizslas at home.
Greg is also the Executive Director of the Center for Medical Technology and Innovation and Associate Professor in the Department of Biomedical Engineering at University of Rochester.













