Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Does my decade old sand bed actually nitrify? Who eats Ammonia in our tanks?
- Thread starter taricha
- Start date
- Tagged users None
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
Not at all really. To correlate these we'd need data and you are right to point out we have almost none.Do we know how a low amount of nitrifiers in a Aquabiomics test correlates to the measured ammonia oxidation rate of the aquarium?
We do have a tiny nugget of data from the aquabiomics article where 3 tanks were started with 3 different kinds of rock.
Figure 6: Relative levels of specific groups of microbes with nitrifying activity. Dashed lines indicate the median (50th percentile) level in a recent survey of established reef tanks. (a) Ammonia-oxidizing Bacteria and Archaea. (b) Nitrite-oxidizing Bacteria, NOB. Orange and green lines overlap in (b) because NOB were not detected in these water samples.
Figure 9. Production of nitrite and nitrate by microbial communities in each tank after while ammonia levels were maintained at approximately 1 ppm. Each symbol represents the average of duplicate tanks.
Note that the the correlation is pretty good for live rock B and Dry rock, but live rock A actually was producing nitrite at the time the microbiome testing found essentially no ammonia oxidizing microbes.
Last edited:
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
Interestingly, that's exactly what flampton said here as a way to shift the system towards nitrification if you really wanted to (though he wasn't advocating that)Just me or does anyone else foresee this experiment leading us back to under gravel filer plates and deep sand beds like we used in the 80's to increase efficiency?
Focus on older techniques of removal e.g. socks, skimmers, water changes, lower lighting if possible and use high protein frozen foods. Maybe use a cryptic refugium as well to help remove excess DOC?? There is also that reverse undergravel filter idea out there. I assume that would keep oxygenation up throughout the bed.
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
OK, @AquaBiomics Data
At the time I collected this sample to go to aquabiomics, I measured my sandbed nitrification rate.
My sand was nitrifying ~0.33 ppm Total Ammonia-Nitrogen per day. Details in post 165
My system overall was consuming ammonia at a max rate of ~2.8 ppm TAN per day (0.115 per hour)
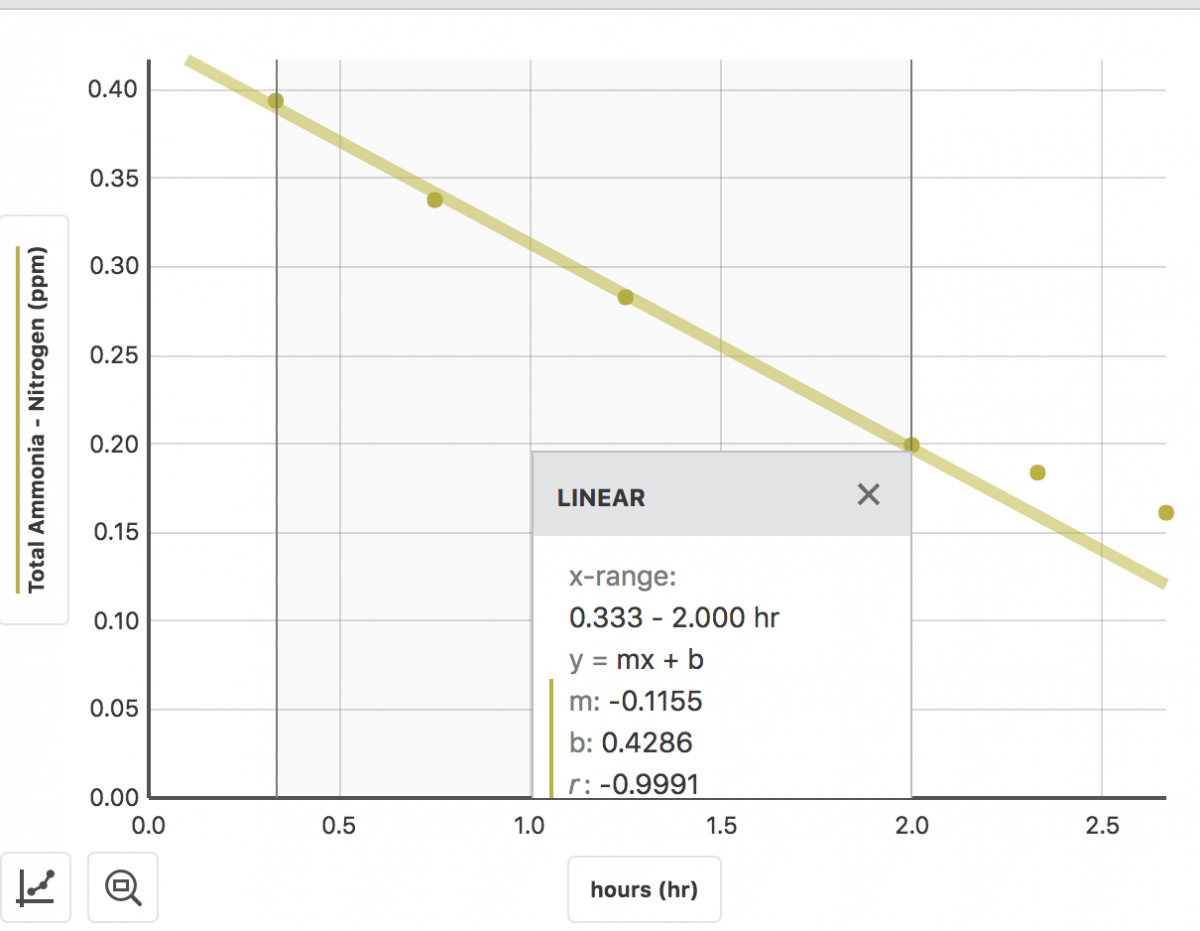
So my sand nitrification was was accounting for only 0.33/2.8 or 12% of my tank's total ammonia consumption. So what does my proportion of nitrifiers look like according to aquabiomics???

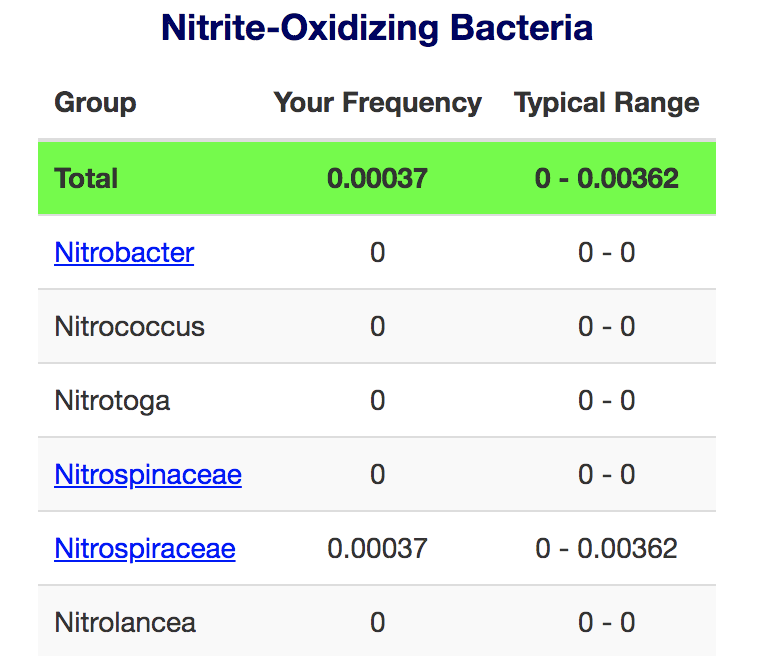
(Complete report attached as pdf)
So both Ammonia oxidizers and nitrite oxidizers are above 50th percentile, meaning that more sequences from these groups were detected in my tank than over half of the tanks tested!
Ammonia oxidizers show both bacteria and archaea - with archaea dominating the count.
So what to make of this?
If we take my data + aquabiomics data at face value, it would point to the conclusion that my system with its low sand nitrifying rate compared to a large ammonia uptake rate for the system is not an anomaly, but is the case across the majority of tanks in the hobby. Ammonia nitrification may only be a bit player in the average established hobby system.
But if you don't like that conclusion, there are plenty of others to choose from
There are a million reasons this isn't a solid leap to make. As @Dan_P suggested, we have no idea how the genetic sequences correlate to chemistry measurements, we have like 2 data points on that.
Some people like @MnFish1 argue that aquabiomics finding wild variation in how many nitrifiers hobby systems have, actually just reflect weakness in the sampling methods.
I lean toward the conclusion that nitrification is a bit player in the average system hobby-wide, and that the aquabiomics data provides some support for that.
( I do have a bone to pick with the aquabiomics data regarding the archaea something doesn't add up, but I'll save that for another post.)
At the time I collected this sample to go to aquabiomics, I measured my sandbed nitrification rate.
My sand was nitrifying ~0.33 ppm Total Ammonia-Nitrogen per day. Details in post 165
My system overall was consuming ammonia at a max rate of ~2.8 ppm TAN per day (0.115 per hour)
So my sand nitrification was was accounting for only 0.33/2.8 or 12% of my tank's total ammonia consumption. So what does my proportion of nitrifiers look like according to aquabiomics???
(Complete report attached as pdf)
So both Ammonia oxidizers and nitrite oxidizers are above 50th percentile, meaning that more sequences from these groups were detected in my tank than over half of the tanks tested!
Ammonia oxidizers show both bacteria and archaea - with archaea dominating the count.
So what to make of this?
If we take my data + aquabiomics data at face value, it would point to the conclusion that my system with its low sand nitrifying rate compared to a large ammonia uptake rate for the system is not an anomaly, but is the case across the majority of tanks in the hobby. Ammonia nitrification may only be a bit player in the average established hobby system.
But if you don't like that conclusion, there are plenty of others to choose from
There are a million reasons this isn't a solid leap to make. As @Dan_P suggested, we have no idea how the genetic sequences correlate to chemistry measurements, we have like 2 data points on that.
Some people like @MnFish1 argue that aquabiomics finding wild variation in how many nitrifiers hobby systems have, actually just reflect weakness in the sampling methods.
I lean toward the conclusion that nitrification is a bit player in the average system hobby-wide, and that the aquabiomics data provides some support for that.
( I do have a bone to pick with the aquabiomics data regarding the archaea something doesn't add up, but I'll save that for another post.)
Attachments
- Joined
- Dec 28, 2016
- Messages
- 22,730
- Reaction score
- 21,905
I do not see where the data you present suggests that nitrification is a bit-player in the average system hobby-wide. Unless you've seen the actual curve. It would be nice to know not just the 'percentile' above or below, it would be nice to also know the mean and standard deviation for each bacteria. i.e. is the standard deviation is 'high', then most tanks will fall in what would be considered a 'normal range' - right?Some people like @MnFish1 argue that aquabiomics finding wild variation in how many nitrifiers hobby systems have, actually just reflect weakness in the sampling methods.
I lean toward the conclusion that nitrification is a bit player in the average system hobby-wide, and that the aquabiomics data provides some support for that.
If!?Still it works both in waste water treatments and aquarium if it is the right organisms
Sincerely Lasse
How many different organisms are you talking about. Which are the right organisms ? How the right organisms stay alive in a closed bottle? Chemo-autotrophic nitrifiers? How they will retrieve their energy to stay active when in a closed bottle?
What is in the bottle? Exactly!
One can not maintain a nitrifying biofilm in a bottle. Even if it would be possiple to deliver archaea and chemo-autotrophic nitrifiers in a bottle, it would make little difference because it would take the time needed for all bacteria needed to build up a community and interconnect.
Heterotrophic nitrifiers may grow fast if enough organic carbon and building materials are available but the heterotropic carrying capacity will be lost when organic carbon becomes limited available. A high heterotrophic carying capacity will prevent the installation of a normal balanced carrying capacity, causing problems when you the least expect as there is no previous warning for an insufficient carrying capacity.
Do you want an aquarium with a carrying capacity based on heterotrophs? Do you want to mange a live support system as it was a wastewater plant?
I think they lower the pH in the bottle which makes them go dormant, vinegar or something.If!?
How many different organisms are you talking about. Which are the right organisms ? How the right organisms stay alive in a closed bottle? Chemo-autotrophic nitrifiers? How they will retrieve their energy to stay active when in a closed bottle?
What is in the bottle? Exactly!
One can not maintain a nitrifying biofilm in a bottle. Even if it would be possiple to deliver archaea and chemo-autotrophic nitrifiers in a bottle, it would make little difference because it would take the time needed for all bacteria needed to build up a community and interconnect.
Heterotrophic nitrifiers may grow fast if enough organic carbon and building materials are available but the heterotropic carrying capacity will be lost when organic carbon becomes limited available. A high heterotrophic carying capacity will prevent the installation of a normal balanced carrying capacity, causing problems when you the least expect as there is no previous warning for an insufficient carrying capacity.
Do you want an aquarium with a carrying capacity based on heterotrophs? Do you want to mange a live support system as it was a wastewater plant?
- Joined
- Sep 21, 2018
- Messages
- 6,654
- Reaction score
- 7,142
These two data points seem to call into question a simple connection between knowing the bacteria and predicting the population‘s metabolism. Where is the disconnect? My guess right now is that what DNA we collect does accurately reflect the organisms’ metabolism we are monitoring. We may need some very clever experiments and observations to sort this out. I am thinking that I should get the ten test discount from Aquobiomics and help increase the number of our our data points.Not at all really. To correlate these we'd need data and you are right to point out we have almost none.
We do have a tiny nugget of data from the aquabiomics article where 3 tanks were started with 3 different kinds of rock.
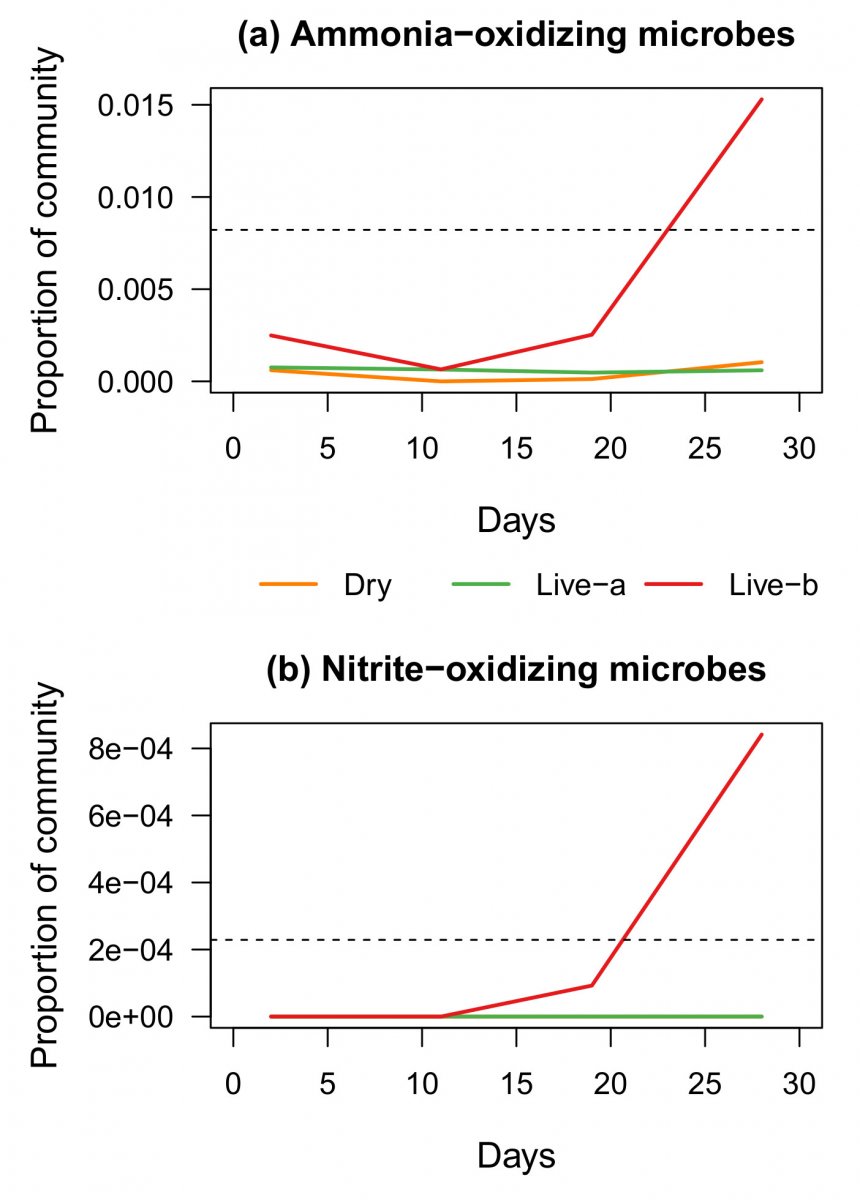
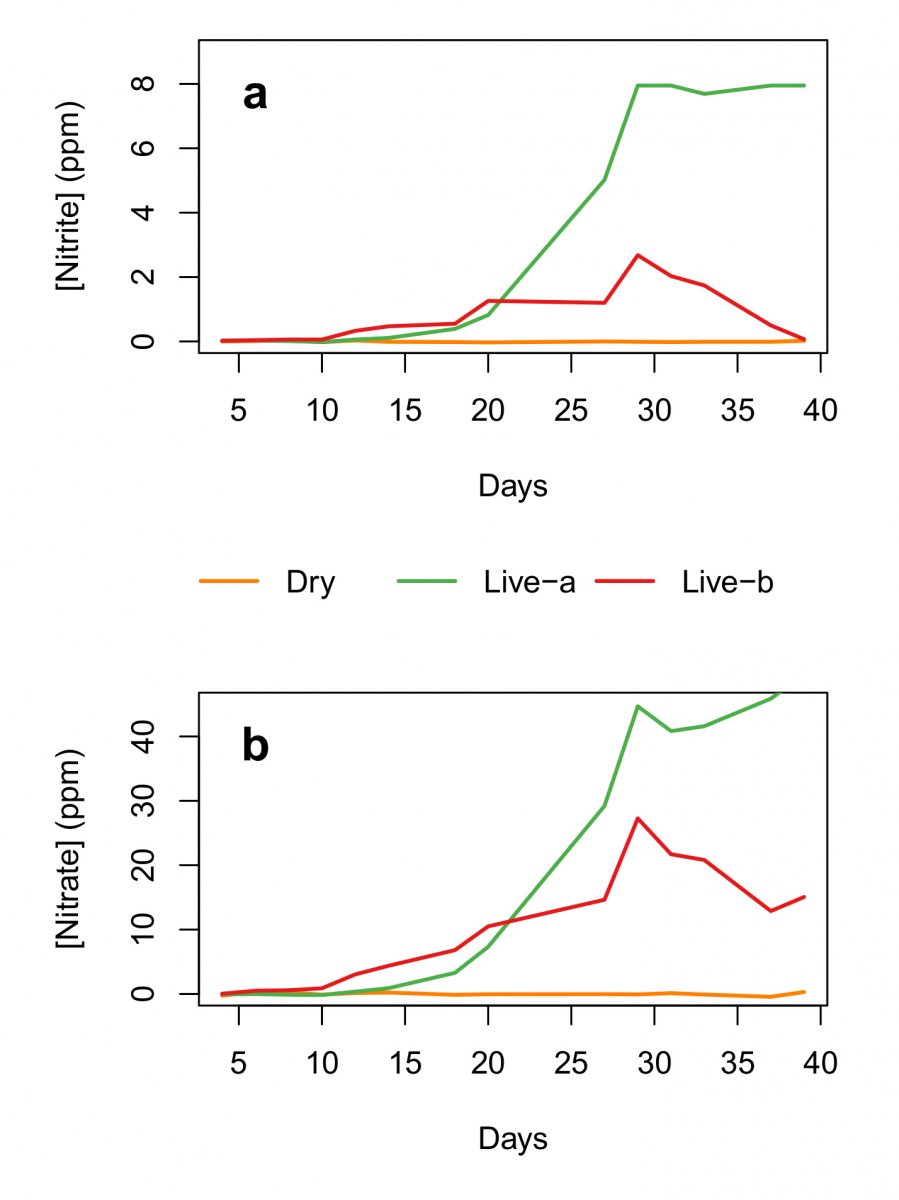
Note that the the correlation is pretty good for live rock B and Dry rock, but live rock A actually was producing nitrite at the time the microbiome testing found essentially no ammonia oxidizing microbes.
- Joined
- Sep 21, 2018
- Messages
- 6,654
- Reaction score
- 7,142
Just thought of something funny. Here you are working hard to understand the big picture of the entire universe of the aquarium while I am off in the corner being totally frustrated trying to grow a nitrifying biofilm on a glass slide. In terms of scale, little bit like cosmology vs the quantum mechanics.Not at all really. To correlate these we'd need data and you are right to point out we have almost none.
We do have a tiny nugget of data from the aquabiomics article where 3 tanks were started with 3 different kinds of rock.


Note that the the correlation is pretty good for live rock B and Dry rock, but live rock A actually was producing nitrite at the time the microbiome testing found essentially no ammonia oxidizing microbes.
- Joined
- Dec 28, 2016
- Messages
- 22,730
- Reaction score
- 21,905
According to at least one company - (who could be lying) - which would be up to you to prove. They ship bacteria cold - with a couple (several?) month expiration date. They are not 'chemo-autotrophs' according to their literature. But - that is how the 'right' organisms stay alive in a closed bottle. How do obligate autotrophs stay alive in a dormant tank (its easy - they go dormant). Its widely known.If!?
How many different organisms are you talking about. Which are the right organisms ? How the right organisms stay alive in a closed bottle? Chemo-autotrophic nitrifiers? How they will retrieve their energy to stay active when in a closed bottle?
What is in the bottle? Exactly!
One can not maintain a nitrifying biofilm in a bottle. Even if it would be possiple to deliver archaea and chemo-autotrophic nitrifiers in a bottle, it would make little difference because it would take the time needed for all bacteria needed to build up a community and interconnect.
Heterotrophic nitrifiers may grow fast if enough organic carbon and building materials are available but the heterotropic carrying capacity will be lost when organic carbon becomes limited available. A high heterotrophic carying capacity will prevent the installation of a normal balanced carrying capacity, causing problems when you the least expect as there is no previous warning for an insufficient carrying capacity.
Do you want an aquarium with a carrying capacity based on heterotrophs? Do you want to mange a live support system as it was a wastewater plant?
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
I'm not making a very fancy argument. I don't think I need to see the curve for a measure to know that if something is above 50th percentile, then the majority are less than that.I do not see where the data you present suggests that nitrification is a bit-player in the average system hobby-wide. Unless you've seen the actual curve. It would be nice to know not just the 'percentile' above or below, it would be nice to also know the mean and standard deviation for each bacteria. i.e. is the standard deviation is 'high', then most tanks will fall in what would be considered a 'normal range' - right?
I'm saying nitrification is a minor player (for ammonia) in my system because the rate of nitrification is on the order of 1/10 of the rate that the system overall can process ammonia. That is to say ammonia assimilation >> nitrification. And I'm saying this case seems widespread in the hobby because my detected nitrifier frequency (ammonia and nitrite oxidizers) was greater than at least half of the tanks they've tested. >50 percentile is relevant here.
I haven't seen the curve but I know we aren't talking about small variations in a near constant measure.
this is a hint of what a few points on the curve may look like.
EM: One of the things that really stood out about your tank for me is the high levels of nutrient-processing microbes. Ammonia-oxidizing microbes made up almost 8% of your sample, which is higher than about 85% of aquariums I’ve tested, about twice as high as the average sample. Your sample also had high levels of nitrite-oxidizing bacteria (0.9%), which is among the highest of any tanks tested (higher than 96% of samples), and over 3-times higher than the average sample.
This may not come as a surprise, since we expect to find these in a tank with a functional biofilter. But in fact, many established tanks have very low levels of these microbes. In some cases, nitrite-oxidizing bacteria aren’t even detected (although they’re likely still present at low levels).
I think although detected less, the NOB might be a better anchor point for my argument. The entire existence of this group is dependent on the oxidation of ammonia in the first place.
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
For those worried about what's in the bottles, whether these are chemoautotrophs or heterotrophs...
Biospira says right on the label: "Patented mix of nitrococcus, nitrosomonas, nitrosospira and nitrospira"
I posted earlier in this thread how a normal 9 day cycle of feeding them nothing but ammonia, they developed a processing rate of ~1.8ppm Total ammonia Nitrogen / day.
In a subsequent post, I showed this capability was almost all in the sand and not the water. Biofilm is doing the work.
The bottle bacteria, myth or fact thread demonstrated conclusively that multiple products: Biospira, One and Only, and Fritz Turbostart need zero Carbon added to process ammonia. Yet it was also showed that many other products couldn't touch ammonia until you fed them carbon (fish flakes) first, these are the heterotrophs.
Dr. Tim also explained to Belgian Anthias directly how nitrifiers can be sustained in a bottle, no ammonia - low activity, they wait with little energy cost. Somewhere I saw a paper posted here (it might have been by Dr Tim) about nitrifiers becoming active again after years with zero ammonia.
I could do an aquabiomics sample of a dry rock tank cycled with just biospira, but I think those that could be convinced already are.
Biospira says right on the label: "Patented mix of nitrococcus, nitrosomonas, nitrosospira and nitrospira"
I posted earlier in this thread how a normal 9 day cycle of feeding them nothing but ammonia, they developed a processing rate of ~1.8ppm Total ammonia Nitrogen / day.
In a subsequent post, I showed this capability was almost all in the sand and not the water. Biofilm is doing the work.
The bottle bacteria, myth or fact thread demonstrated conclusively that multiple products: Biospira, One and Only, and Fritz Turbostart need zero Carbon added to process ammonia. Yet it was also showed that many other products couldn't touch ammonia until you fed them carbon (fish flakes) first, these are the heterotrophs.
Dr. Tim also explained to Belgian Anthias directly how nitrifiers can be sustained in a bottle, no ammonia - low activity, they wait with little energy cost. Somewhere I saw a paper posted here (it might have been by Dr Tim) about nitrifiers becoming active again after years with zero ammonia.
I could do an aquabiomics sample of a dry rock tank cycled with just biospira, but I think those that could be convinced already are.
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
Hah. My questions are usually too broad, so even when I get answers, I don't really understand them.Here you are working hard to understand the big picture of the entire universe of the aquarium while I am off in the corner being totally frustrated trying to grow a nitrifying biofilm on a glass slide.
- Joined
- Dec 28, 2016
- Messages
- 22,730
- Reaction score
- 21,905
THANKS - EXACTLY - This is totally correct. One poster has an agenda (one that likes anthias)For those worried about what's in the bottles, whether these are chemoautotrophs or heterotrophs...
Biospira says right on the label: "Patented mix of nitrococcus, nitrosomonas, nitrosospira and nitrospira"
I posted earlier in this thread how a normal 9 day cycle of feeding them nothing but ammonia, they developed a processing rate of ~1.8ppm Total ammonia Nitrogen / day.
In a subsequent post, I showed this capability was almost all in the sand and not the water. Biofilm is doing the work.
The bottle bacteria, myth or fact thread demonstrated conclusively that multiple products: Biospira, One and Only, and Fritz Turbostart need zero Carbon added to process ammonia. Yet it was also showed that many other products couldn't touch ammonia until you fed them carbon (fish flakes) first, these are the heterotrophs.
Dr. Tim also explained to Belgian Anthias directly how nitrifiers can be sustained in a bottle, no ammonia - low activity, they wait with little energy cost. Somewhere I saw a paper posted here (it might have been by Dr Tim) about nitrifiers becoming active again after years with zero ammonia.
I could do an aquabiomics sample of a dry rock tank cycled with just biospira, but I think those that could be convinced already are.
- Joined
- Dec 28, 2016
- Messages
- 22,730
- Reaction score
- 21,905
You're right - I do not trust the samples - I would suggest if you sampled 10 areas from your tank - you would get multiple different answers to your question. Completely mix up the water/sand/etc - in your tank - until its cloudy - then sample it. See what it shows - it will be completely differentI'm not making a very fancy argument. I don't think I need to see the curve for a measure to know that if something is above 50th percentile, then the majority are less than that.
I'm saying nitrification is a minor player (for ammonia) in my system because the rate of nitrification is on the order of 1/10 of the rate that the system overall can process ammonia. That is to say ammonia assimilation >> nitrification. And I'm saying this case seems widespread in the hobby because my detected nitrifier frequency (ammonia and nitrite oxidizers) was greater than at least half of the tanks they've tested. >50 percentile is relevant here.
I haven't seen the curve but I know we aren't talking about small variations in a near constant measure.
this is a hint of what a few points on the curve may look like.
I think although detected less, the NOB might be a better anchor point for my argument. The entire existence of this group is dependent on the oxidation of ammonia in the first place.
I thought I did not need to state that I meant AOO and NOO because the thread is about these organisms. They ate mostly Chemoautotropic organism - if not all. I do not know how they get these organisms dormant - but the can clearly go dormant. I have used these products to seed nitrification filter since the 80:ties. In nature - these organism usually are found in the upper layer of the soils - as all understand - ammonia and nitrite is not a steady source in the soil - it vary among seasons and input of NH3/NH4. Hence their activity vary between 0 and full activity. I know that you know about sequential batch reactors there you vary the environment from aerobic to anaerobic environment. The AOO and NOO survive the anaerobic phase and have full function when it become aerobic again.If!?
How many different organisms are you talking about. Which are the right organisms ? How the right organisms stay alive in a closed bottle? Chemo-autotrophic nitrifiers? How they will retrieve their energy to stay active when in a closed bottle?
What is in the bottle? Exactly!
By the way - the difference between a waste water plant and and fully working aquarium is only the load of organic and inorganic compounds - the principles is the same.
Sincerely Lasse
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
Here's my issue. It's a bit of a paradox.I do have a bone to pick with the aquabiomics data regarding the archaea. Something doesn't add up, but I'll save that for another post.
1) I measure almost no nitrification in my water, and I easily measure nitrification in my sand.
2) Aquabiomics finds that the vast majority of Ammonia oxidizing microbe sequences in my sample are archaea with only a tiny slice being bacteria.
3) microbiology folks say that archaea are active mostly in the water...
But in fact by far the most abundant ammonia-oxidizer in saltwater aquariums are not Bacteria, but rather Archaea. In this case they are free-living Archaea that live in the water column. So the often repeated statement that nitrifiers don't live in the water is not strictly true. In fact, in terms of cell numbers, thats probably where most of them are. But many types (especially nitrite-oxidizing bacteria) are much more abundant in the biofilm.
I'm not sure what the resolution is to the paradox.
Could simply be that the notion about their activity in the water is incorrect and that our saltwater archaea are active on surfaces as much or more. (It sounds flip to say that maybe the people who study archaea are wrong, but from what I gather, they are mostly studied by picking up their genes and enzymes in the environment. rarely cultured.)
Or it could be that they are a tiny population of cells that sheds a lot of genetic sequences in the water where they are easy to collect (vs biofilm), or maybe the cells themselves have a tiny appetite for ammonia compared to a cell of ammonia oxidizing bacteria.
if it's either of the last two, then it goes to Dan's point below. The correlation between detected sequences and metabolism of ammonia would be pretty weak. Some of this ought to be answered in published papers, I think.
That study was done early. I wonder if it was before aquabiomics switched to adding a biofilm swab as a standard part of the test. "swab inside a dark contiinuously submerged pipe." Also, it's worth pointing out aquabiomics isn't measuring live cells, or even cells at all - just genetic sequences.These two data points seem to call into question a simple connection between knowing the bacteria and predicting the population‘s metabolism. Where is the disconnect? My guess right now is that what DNA we collect doesn't accurately reflect the organisms’ metabolism we are monitoring. We may need some very clever experiments and observations to sort this out. I am thinking that I should get the ten test discount from Aquobiomics and help increase the number of our our data points.
All of this gets at some of @MnFish1 skepticism as well. Maybe the test protocol just isn't a good way to measure your nitrifier population.
- Joined
- May 22, 2016
- Messages
- 6,526
- Reaction score
- 10,060
Partial answer to your question. Turns out that this was actually the case...These two data points seem to call into question a simple connection between knowing the bacteria and predicting the population‘s metabolism. Where is the disconnect?
That study was done early. I wonder if it was before aquabiomics switched to adding a biofilm swab as a standard part of the test. "swab inside a dark continuously submerged pipe."
Here, he addresses the fact that the low quality live rock (Live Rock A) have a discrepancy between chemistry and the genetic info.
So yeah, that data was without biofilm sampling that is now standard.This comparison also serves to illustrate a point made previously that undetected microbes are not necessarily absent. The chemistry data clearly show that live rock a contained some NOB, even though they were present at low levels below the limit of detection for DNA sequencing of water samples. Direct comparison of water and biofilm samples from a single tank confirmed that these microbes can be absent in the water column but present in biofilm. Based on these and related observations, we’re including both biofilm and water samples in all current and future testing, to increase our ability to detect these important microbial components of the biological filter.
Never heard that - always heard that it is in a biofilmmicrobiology folks say that archaea are active mostly in the water...
Sincerely Lasse
I´m not sure that this is the best place to take sample from. If I do the test again - I will swab in the sand insteadThat study was done early. I wonder if it was before aquabiomics switched to adding a biofilm swab as a standard part of the test. "swab inside a dark contiinuously submerged pipe."
Sincerely Lasse
Similar threads
- Replies
- 38
- Views
- 678
- Replies
- 1
- Views
- 77
- Replies
- 4
- Views
- 160
New Posts
-
-
-
How long should i mix instant ocean salt for?
- Latest: EeyoreIsMySpiritAnimal
-