Dear Reef2Reefers!
I have seen a lot of skepticism regarding seawater laboratory analysis in this forum, especially when in comes to reported values for trace elements. I must say, that all skepticism is very well justified, since there is many potential pitfalls in ICP analysis: Sampling, sample handling in the laboratory, laboratory contamination, interferences, wrong interpretation of data, and many more. Those points even increase in importance when very low detection limits are crucial.
Disclaimer: I can speak here only for the procedures in our lab (Oceamo). I hope this post does not have too much of an advertisment character (@Randy Holmes-Farley, please remove it if this should be the case)
For transparency reasons and to build some confidence in trace element analysis id like to share some data with you. We have started our ICP-MS service late last year. ICP-MS is way more sensitive, so we can detect and quantify trace elements and pollutants at much lower concentration compared to ICP-OES - but ICP-MS also comes with many challenges the analytical chemist must handle to provide reliable results.
To control the quality of our data we are using the seawater certified reference material ERM-CA403 supplied by the European Comission. This is seawater, that has very clearly determined levels of certain trace elements. Im Posting the certificate of analysis here:

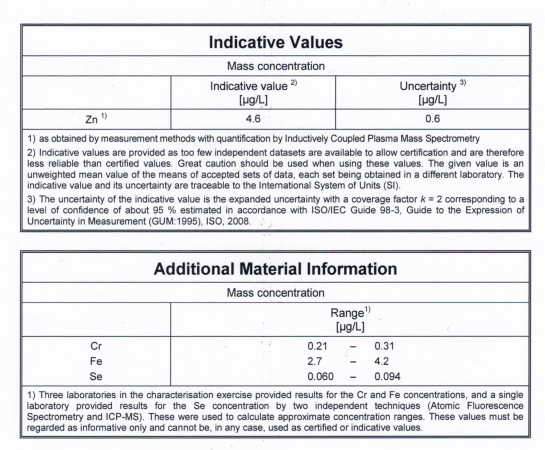
This reference material is included in all our ICP-MS sequences, to check if our measurements are correct.
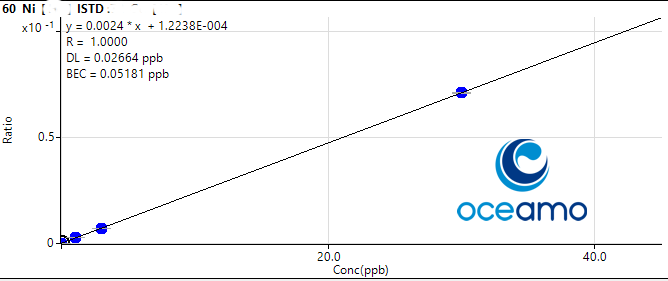
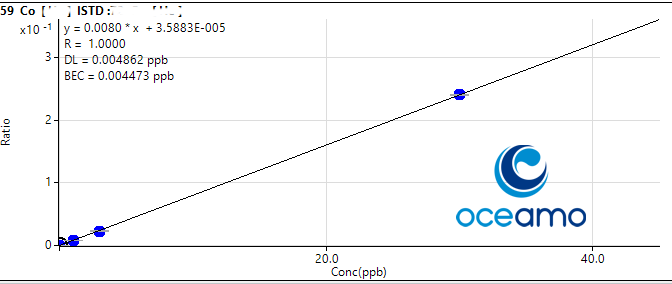
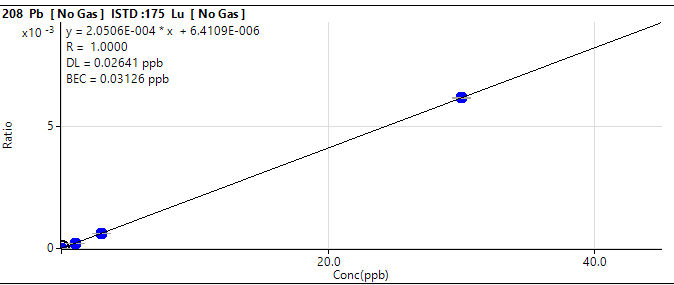
That you get a feeling for the level of accuracy and uncertainty you can expect, i am posting our results and the certified value for the more difficult elements here, and will also supply the calibration plots that show linearity and our limit of detection.
Lets start with Nickel:
Concentration in ERM-CA403: 1,04 µg/l (uncertainty 0,16 µg/l)
Oceamo ICP-MS measurement 1: 1,15 µg/l
Oceamo ICP-MS measurement 2: 1,17 µg/l
Cobalt:
Concentration in ERM-CA403: 0,074 µg/l (uncertainty 0,011 µg/l)
Oceamo ICP-MS measurement 1: 0,063 µg/l
Oceamo ICP-MS measurement 2: 0,068 µg/l
Copper:
Concentration in ERM-CA403: 0,87 µg/l (uncertainty 0,13 µg/l)
Oceamo ICP-MS measurement 1: 0,754 µg/l
Oceamo ICP-MS measurement 2: 0,871 µg/l
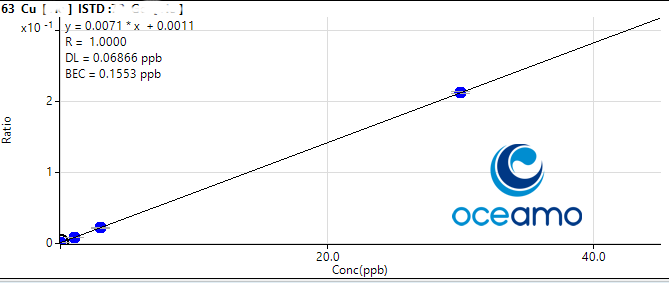
Cadmium:
Concentration in ERM-CA403: 0,094 µg/l (uncertainty 0,011 µg/l)
Oceamo ICP-MS measurement 1: 0,094 µg/l
Oceamo ICP-MS measurement 2: 0,099 µg/l
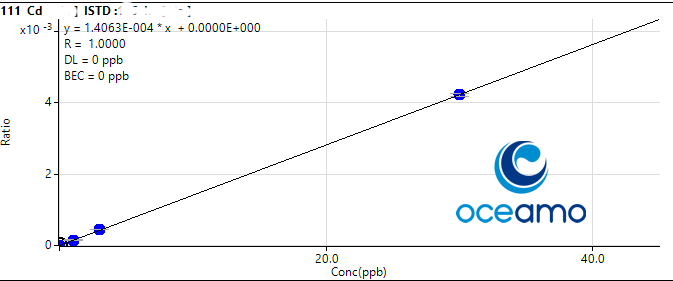
Arsenic:
Concentration in ERM-CA403: 1,90 µg/l (uncertainty 0,13 µg/l)
Oceamo ICP-MS measurement 1: 1,86 µg/l
Oceamo ICP-MS measurement 2: 1,63 µg/l
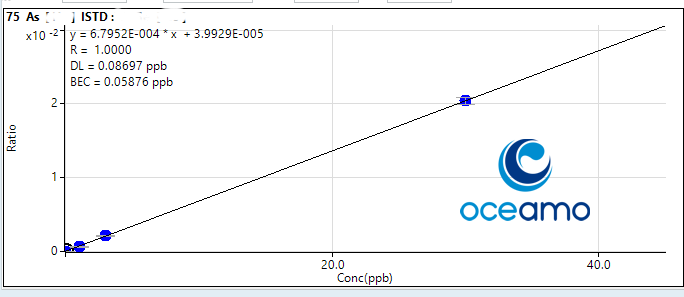
Selenium:
Concentration in ERM-CA403: 0,06-0,094 µg/l
Oceamo ICP-MS measurement 1: 0,083 µg/l
Oceamo ICP-MS measurement 2: 0,092 µg/l
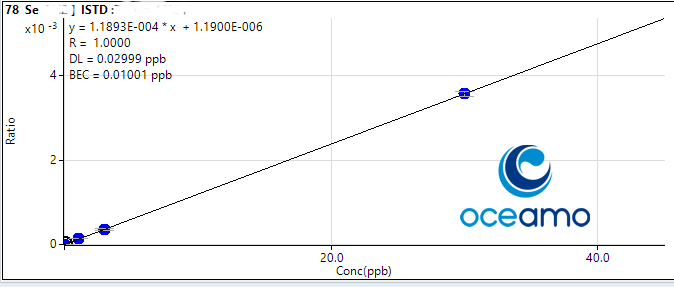
Lead:
Concentration in ERM-CA403: 0,098 µg/l (uncertainty 0,010 µg/l)
Oceamo ICP-MS measurement 1: 0,114 µg/l
Oceamo ICP-MS measurement 2: 0,097 µg/l
I hope you found this information useful, and that it contributes to lift the curtains from the blackbox "seawater laboratory".
By this data you can also see, why i have decided to expand towards ICP-MS: The levels measured here are all within the relevant concentration range of those trace elements in seawater (and thus also in our aquaria). - But are at the same time below the realistic limit of detection for ICP-OES, and thus would appear on the analysis report as "non detectable". ICP-OES (which we also frequently use) is a great tool for many elements that appear in higher concentration ranges, and to (in many cases) detect elevated levels - but fails to reliably diagnose (too) low levels for many essential trace elements. This is where ICP-MS fills the void.
If youre having any questions i am happy to help!
Best regards,
Christoph
I have seen a lot of skepticism regarding seawater laboratory analysis in this forum, especially when in comes to reported values for trace elements. I must say, that all skepticism is very well justified, since there is many potential pitfalls in ICP analysis: Sampling, sample handling in the laboratory, laboratory contamination, interferences, wrong interpretation of data, and many more. Those points even increase in importance when very low detection limits are crucial.
Disclaimer: I can speak here only for the procedures in our lab (Oceamo). I hope this post does not have too much of an advertisment character (@Randy Holmes-Farley, please remove it if this should be the case)
For transparency reasons and to build some confidence in trace element analysis id like to share some data with you. We have started our ICP-MS service late last year. ICP-MS is way more sensitive, so we can detect and quantify trace elements and pollutants at much lower concentration compared to ICP-OES - but ICP-MS also comes with many challenges the analytical chemist must handle to provide reliable results.
To control the quality of our data we are using the seawater certified reference material ERM-CA403 supplied by the European Comission. This is seawater, that has very clearly determined levels of certain trace elements. Im Posting the certificate of analysis here:
This reference material is included in all our ICP-MS sequences, to check if our measurements are correct.
That you get a feeling for the level of accuracy and uncertainty you can expect, i am posting our results and the certified value for the more difficult elements here, and will also supply the calibration plots that show linearity and our limit of detection.
Lets start with Nickel:
Concentration in ERM-CA403: 1,04 µg/l (uncertainty 0,16 µg/l)
Oceamo ICP-MS measurement 1: 1,15 µg/l
Oceamo ICP-MS measurement 2: 1,17 µg/l

Cobalt:
Concentration in ERM-CA403: 0,074 µg/l (uncertainty 0,011 µg/l)
Oceamo ICP-MS measurement 1: 0,063 µg/l
Oceamo ICP-MS measurement 2: 0,068 µg/l

Copper:
Concentration in ERM-CA403: 0,87 µg/l (uncertainty 0,13 µg/l)
Oceamo ICP-MS measurement 1: 0,754 µg/l
Oceamo ICP-MS measurement 2: 0,871 µg/l

Cadmium:
Concentration in ERM-CA403: 0,094 µg/l (uncertainty 0,011 µg/l)
Oceamo ICP-MS measurement 1: 0,094 µg/l
Oceamo ICP-MS measurement 2: 0,099 µg/l

Arsenic:
Concentration in ERM-CA403: 1,90 µg/l (uncertainty 0,13 µg/l)
Oceamo ICP-MS measurement 1: 1,86 µg/l
Oceamo ICP-MS measurement 2: 1,63 µg/l

Selenium:
Concentration in ERM-CA403: 0,06-0,094 µg/l
Oceamo ICP-MS measurement 1: 0,083 µg/l
Oceamo ICP-MS measurement 2: 0,092 µg/l

Lead:
Concentration in ERM-CA403: 0,098 µg/l (uncertainty 0,010 µg/l)
Oceamo ICP-MS measurement 1: 0,114 µg/l
Oceamo ICP-MS measurement 2: 0,097 µg/l

I hope you found this information useful, and that it contributes to lift the curtains from the blackbox "seawater laboratory".
By this data you can also see, why i have decided to expand towards ICP-MS: The levels measured here are all within the relevant concentration range of those trace elements in seawater (and thus also in our aquaria). - But are at the same time below the realistic limit of detection for ICP-OES, and thus would appear on the analysis report as "non detectable". ICP-OES (which we also frequently use) is a great tool for many elements that appear in higher concentration ranges, and to (in many cases) detect elevated levels - but fails to reliably diagnose (too) low levels for many essential trace elements. This is where ICP-MS fills the void.
If youre having any questions i am happy to help!
Best regards,
Christoph