- Joined
- Sep 21, 2018
- Messages
- 6,843
- Reaction score
- 7,321
What is the pH of your system?I've been perplexed on how my pico tank ammonia level never seems to drop despite large water changes and adding bacteria. This is an already established tank that was move almost a week ago.
I would assume that doing a 50% water change should have a noticeable impact on levels. I've done enough changes this week to easily change all the water almost twice with no effect on Ammonia levels.
So I investigated further.
Here is the test for the 5G pico for reference:

I tested the RO water I use for my ATO. This is Seapora RO, which tested at 6 TDS, by the way:

So far so good. Now on to the RO water I use to mix my saltwater. This is Culligan RO that tests at 1 TDS:

Okay... on to the mixed saltwater I'm using for my water changes:
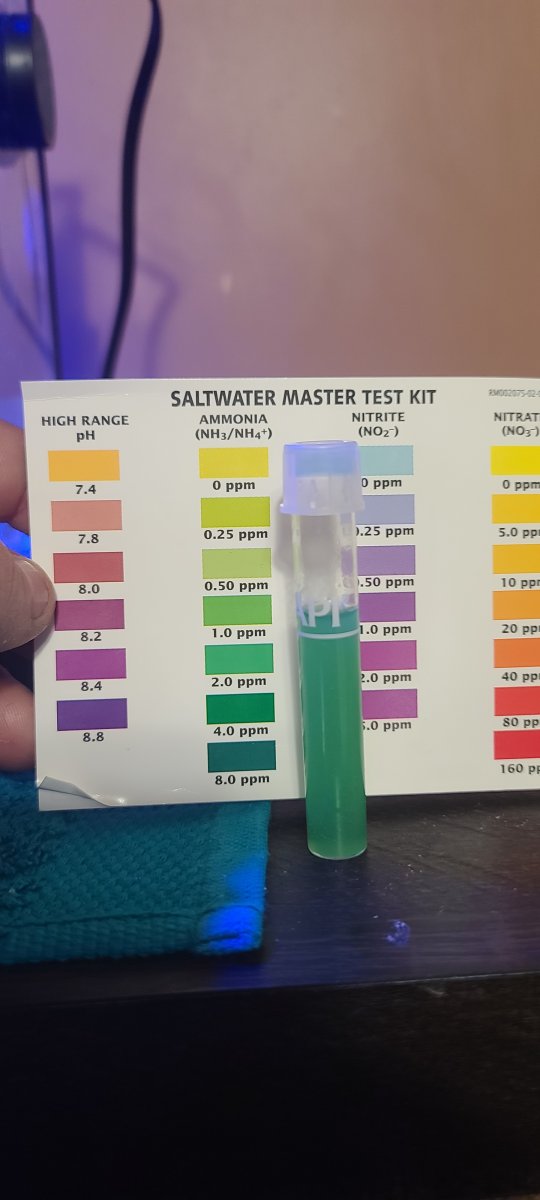
So, I've been doing water changes to drop ammonia by adding ammonia!! What the?! This was mixed in a brand new Brute 20G container that was cleaned and rinse very well before use. The pump used to mix was ran in RO for a few minutes before adding to the fresh RO in the Brute and salt used was Aquavitro Salinity. Could that salt have ammonia?
I have a different salt type, Red Sea I believe, that I will be using to mix up a brand new batch with very soon.
Is there any explanation for this? Does premixed salt "age" and produce ammonia? The bin has always had its lid on when I'm not taking water and there is clearly nothing dead in it.
I'm more confused now than ever.
This is certainly different! Usually an API ammonia test user will be in doubt about the test results being 0 ppm or 0.25 ppm. You have what looks like a positive test for ammonia in your system and salt mix. My Instant Ocean has tenths of a ppm on ammonia and maybe a tenth of a ppm of nitrite. Your test shows a boat load.
I have been thinking about ways the test could be fooled. Maybe a goofy high level of an amino acid would give a false positive.
If your tank inhabitants are fine AND if there is ammonia present, the pH of the system must be low enough to suppress most of the NH4+ from being converted to NH3, the toxic version of the molecule.

















