Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
Trace elements continue to pose a dilemma for reefers who are not sure which chemical forms of which trace elements are needed at what range of concentrations for each individual organism.
We are making some small progress, but have a long way to go.
One of the conundrums that face reefers is that many reef tanks seem to do perfectly fine without measuring or dosing trace elements. Others are fine just adding one (say, iron) or a few in some random element mix made by a company that at best is trying to meet the needs of wide ranging reef tanks with a single product.
How can all of these trace elements be in an optimal range without the high degree of control that some methods using ICP and individual dosing attain?
Here's one way this all works out: the optimal range of concentration may span a vast range from very low to very high.
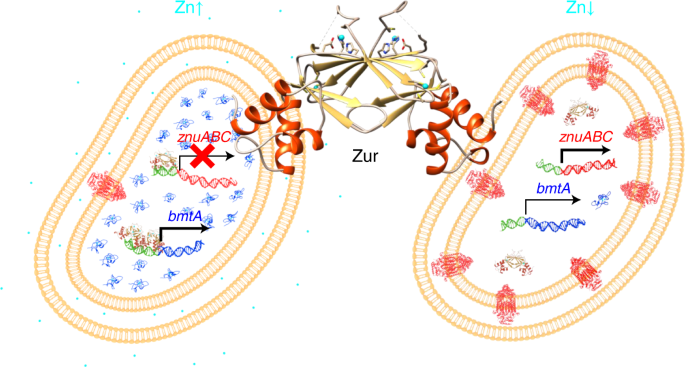
Taking a recent example for a single organism ( Synechococcus cyanobacteria) and a single element (zinc), these folks have shown that the cyano can adapt itself to a huge range of available zinc by having a specific uptake mechanism for it, and which it can ramp up or down as needed when supplies are lowest or highest. This organism is not a priority organism for most reefers, but there's no reason to assume that organisms we care about have not developed similar sorts of mechanisms for their own needs, and we will likely find that many trace elements can vary considerably before being too high or too low for our desired organisms, unlike certain of the tank attributes we currently focus most on (say, calcium or alkalinity).
A single sensor controls large variations in zinc quotas in a marine cyanobacterium
 www.nature.com
www.nature.com
Abstract
Marine cyanobacteria are critical players in global nutrient cycles that crucially depend on trace metals in metalloenzymes, including zinc for CO2 fixation and phosphorus acquisition. How strains proliferating in the vast oligotrophic ocean gyres thrive at ultra-low zinc concentrations is currently unknown. Using Synechococcus sp. WH8102 as a model we show that its zinc-sensor protein Zur differs from all other known bacterial Zur proteins in overall structure and the location of its sensory zinc site. Uniquely, Synechococcus Zur activates metallothionein gene expression, which supports cellular zinc quotas spanning two orders of magnitude. Thus, a single zinc sensor facilitates growth across pico- to micromolar zinc concentrations with the bonus of banking this precious resource. The resultant ability to grow well at both ultra-low and excess zinc, together with overall lower zinc requirements, likely contribute to the broad ecological distribution of Synechococcus across the global oceans.
We are making some small progress, but have a long way to go.
One of the conundrums that face reefers is that many reef tanks seem to do perfectly fine without measuring or dosing trace elements. Others are fine just adding one (say, iron) or a few in some random element mix made by a company that at best is trying to meet the needs of wide ranging reef tanks with a single product.
How can all of these trace elements be in an optimal range without the high degree of control that some methods using ICP and individual dosing attain?
Here's one way this all works out: the optimal range of concentration may span a vast range from very low to very high.
Taking a recent example for a single organism ( Synechococcus cyanobacteria) and a single element (zinc), these folks have shown that the cyano can adapt itself to a huge range of available zinc by having a specific uptake mechanism for it, and which it can ramp up or down as needed when supplies are lowest or highest. This organism is not a priority organism for most reefers, but there's no reason to assume that organisms we care about have not developed similar sorts of mechanisms for their own needs, and we will likely find that many trace elements can vary considerably before being too high or too low for our desired organisms, unlike certain of the tank attributes we currently focus most on (say, calcium or alkalinity).
A single sensor controls large variations in zinc quotas in a marine cyanobacterium

A single sensor controls large variations in zinc quotas in a marine cyanobacterium - Nature Chemical Biology
The zinc-sensor protein Zur in a marine cyanobacterium is distinct from those in other bacteria in structure and location of its sensory zinc site, and facilitates growth across a range of zinc concentrations via activation of a metallothionein gene.
Abstract
Marine cyanobacteria are critical players in global nutrient cycles that crucially depend on trace metals in metalloenzymes, including zinc for CO2 fixation and phosphorus acquisition. How strains proliferating in the vast oligotrophic ocean gyres thrive at ultra-low zinc concentrations is currently unknown. Using Synechococcus sp. WH8102 as a model we show that its zinc-sensor protein Zur differs from all other known bacterial Zur proteins in overall structure and the location of its sensory zinc site. Uniquely, Synechococcus Zur activates metallothionein gene expression, which supports cellular zinc quotas spanning two orders of magnitude. Thus, a single zinc sensor facilitates growth across pico- to micromolar zinc concentrations with the bonus of banking this precious resource. The resultant ability to grow well at both ultra-low and excess zinc, together with overall lower zinc requirements, likely contribute to the broad ecological distribution of Synechococcus across the global oceans.




















