I'm not sure why you are starting using CaCO3 in a mix intended to dissolve since it has quite low solubility. If you feel you want carbonate, I think you'd be better off using potassium or sodium carbonate.
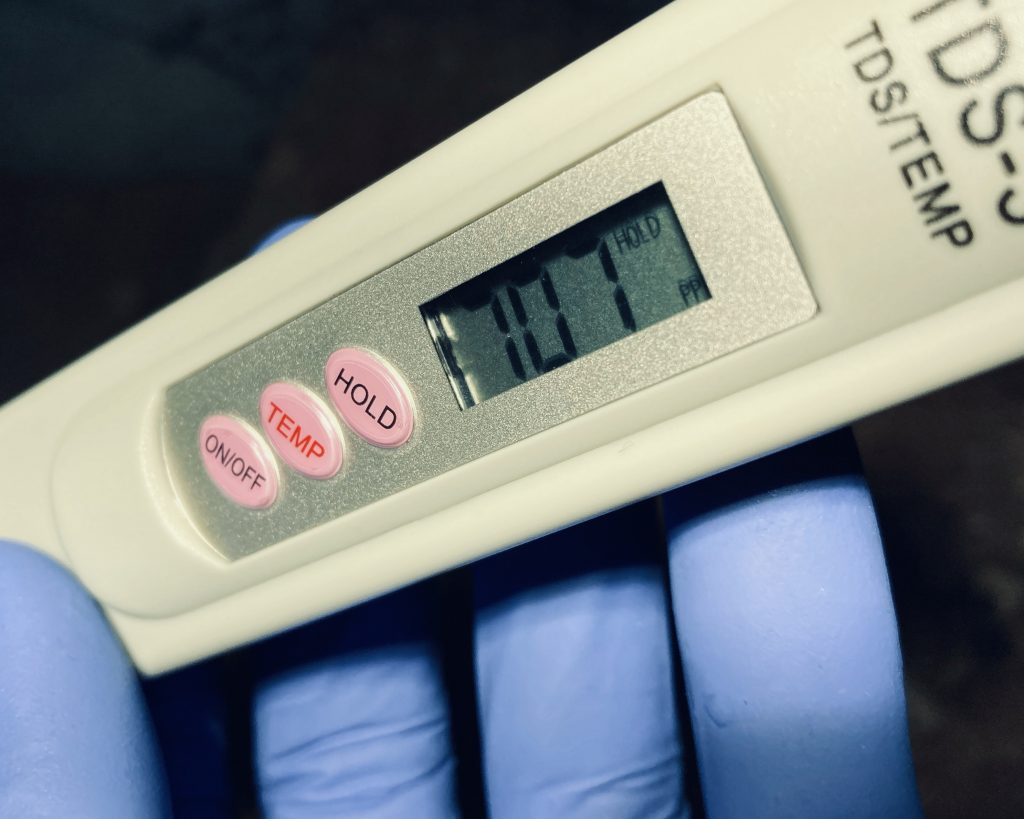
Please see the spreadsheet above. I'm trying to get the ion concentrations close to the target values. LIke I mentioned, this updated blend had very little sediment. Actually, the extra 8-liter batch I have had in this jug for a couple weeks is now completely clear.
If calcium carbonate is near saturation that should advance the goal of getting precipitation on the reef structure rock surfaces where the photosynthetic microorganisms are growing.
Last edited: