- Joined
- Sep 21, 2018
- Messages
- 6,705
- Reaction score
- 7,187
I was thinking about how @taricha did his study, which he reveals below.Could you find it using standards from...say .01 to .04 measured on both...and plot the slope. ..or am I confused
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
I was thinking about how @taricha did his study, which he reveals below.Could you find it using standards from...say .01 to .04 measured on both...and plot the slope. ..or am I confused
What I noticed was that the amount of color required above zero in order to get a reading is more than the color difference error at other concentrations. It requires about 2-3 times the normal uncertainty to get a reading above zero. In other words, they built in a buffer against false positives.
If you want to probe that for your checkers, then use a constant color solution - like drops of tea or koolaid or something, don't bother with the hassle of a chemical test just to check the low range color linearity.
Nearly all analyses I know of is not linear near zero - it is often a error in measurement equipment. High quality gears have compensations to this. To expect this for a equipment that cost $50 - $60 - probably not.
HI-774 and Hi-736 is - IMO - the same meter - it is only the reading thats differ. As mine HI-774 looks like it overestimate my result with 0.08 - IMO - it is probably a fault of individual meters instead of differences between models.
Sincerely Lasse
yes now it is coming back to me! I think I used vanilla at your suggestion to do this with one of the Hanna checkers Thanks for the reminder
Nearly all analyses I know of is not linear near zero - it is often a error in measurement equipment. High quality gears have compensations to this. To expect this for a equipment that cost $50 - $60 - probably not.
Another place you can see this insensitivity near zero / protection against false positives is with mismatched cuvettes.
If you grab random mismatched hanna cuvettes from different test kits and fill them with DI water, you'll probably never get a reading regardless of which you use as the C1 and C2.
But...
When I used a colored solution in C2, and DI water as C1, I got a repeatable difference when I measured the solution in cuvette A to B, vs when I switched the solution to the other cuvette and measured B to A
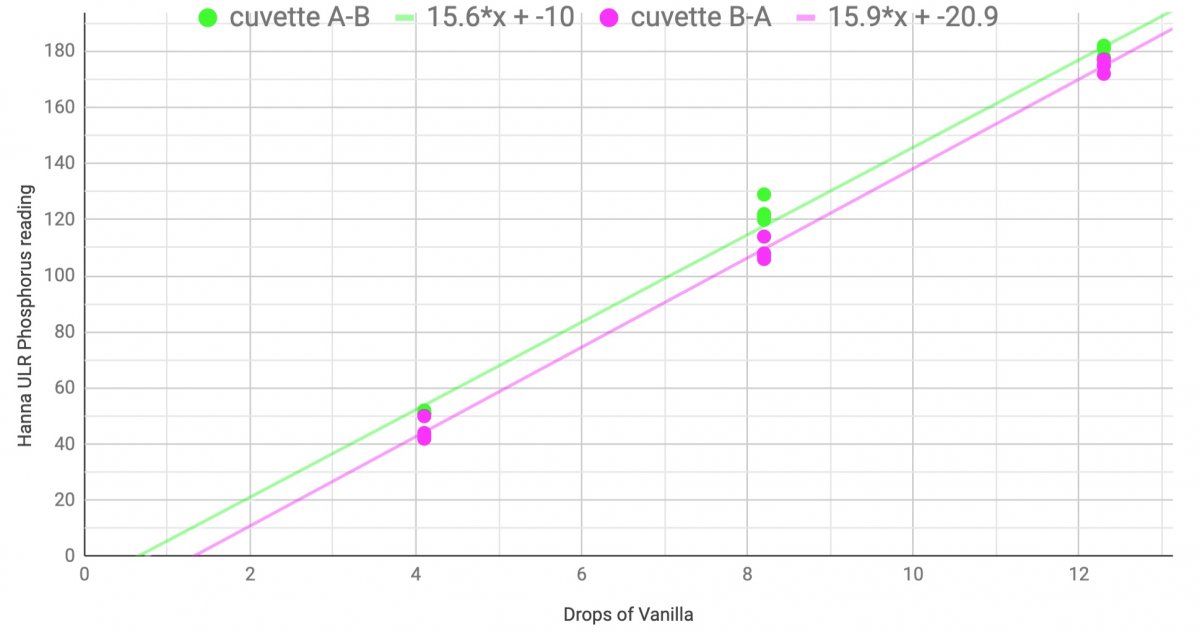
Measurement order changed result by 5 to 10 ppb. But when I fill them both with DI, I don't get a reading of 5 to 10. I get zero repeatably.
I always use two different tubes and zero the first and read zero on the second directly when they are cleaned well. However - your result - IMO - only show the ±5 ppb effect when reading results over 0 - nothing else. but i can misunderstand the graph - or it above my pay level. If the graph does not hit the 0 - - can you trust the measurements?you'll probably never get a reading regardless of which you use as the C1 and C2.
