- Joined
- Aug 5, 2017
- Messages
- 1,493
- Reaction score
- 1,881
No particular opinion on them. I do not have a reason to not trust it.
Randy, that was a double negative
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
No particular opinion on them. I do not have a reason to not trust it.
I don’t have a trident I don’t trust neptune as a company.
I am patiently waiting for a competitor.
I have my $50 Hanna checker that always works.
I have a lot of apex products but I am slowly moving
Away from their products. This is a hobby for me.
I like to keep things simple. I think people complicate this hobby with no need.
My LFS has a show tank in the store. He never test anything
He doesn’t dose any elements or any thing to feed the corals.
The tank is on a calcium reactor / skimmer and that’s it no UV .that tank has some of the best corals I have ever seen and corals eat fish poop and that’s it. Reefers 10yr ago had great tanks you asked them what their alk was they would say I have no idea lol
If you really want confidence in your home test kits make a known "standard" or buy one. Some test kits come with a standard which is great. If you make one yourself make it with a concentration close to that of your reef water. Assuming you prepare your standard correctly it should give you confidence in your results. Titrations for example can be very reliable if you can consistently identify the endpoint and a standard can help refine your technique

Randy, that was a double negative
This is why we can’t have nice things.
Icp test fails are probably less than 10%
But here we are here saying it’s bad.
Somebody reads this thread and decides not to do an icp
Test. A lot of people have been helped fix their tank issues
Because of icp. This is a great tool that we can use and it helps a lot of reefers.
Salt water parameters don’t change in shipping.
Once you takes your sample and put it in a bottle.
And seal it that sample will remain the same.
The same way if you mix a salt water the alk/cal/mag
And trace elements will stay the same in that container for a long time.
BRS did a test and that said the mixed salt water
Tested the same 30 days later.
Icp is not effected by shipping.
More people are getting bad reading from
Their apex triton than icp. But since apex is a sponsor
They get a pass. You spend $600 and when your reagents get low your readings are off. But no one says don’t trust trident.
I find it funny how we question companies that do icp test.
But don’t question BRS test that are conducted by two guys with a camera. But BRS are also sponsors so their
Word is bible.
I am getting ready to send out my icp test.
Good luck with your tanks

This is spot on! I manage 3 water testing laboratories in 2 states. We run multiple ICP, ICPMS, and IC instruments. My labs test my tank water weekly and i do home test kits about once a month. Those advanced pieces of equipment are very reliable but that assumes everything such as calibration and sample prep was done perfectly and that is not always the case. Are those big vendors doing maintenance on their equipment when their Boron calibration starts to drift? I am sure they do but exactly when do they scrap a sample run because the instrument is off spec? Your definition of acceptable may not be the same as theirs. This is why in my business "blind samples" are a common thing. We get water that looks like any other client sample but its a disguised check standard with known concentrations. If we report results for that sample that is not within specs we risk losing our certification. There is no certification for reef water so your results are not being judged by any certified authority.. .just something to keep in mind.
If you really want confidence in your home test kits make a known "standard" or buy one. Some test kits come with a standard which is great. If you make one yourself make it with a concentration close to that of your reef water. Assuming you prepare your standard correctly it should give you confidence in your results. Titrations for example can be very reliable if you can consistently identify the endpoint and a standard can help refine your technique
I disagree with you. I pressed several major ICP companies to show me their validation documents, their calibration protocol, etc. The German companies provided me with the documentation I was hoping for. The American ones that i poked (did not do them all) could not show me these docs. Without validation and calibration, the numbers they spit out are worthless.
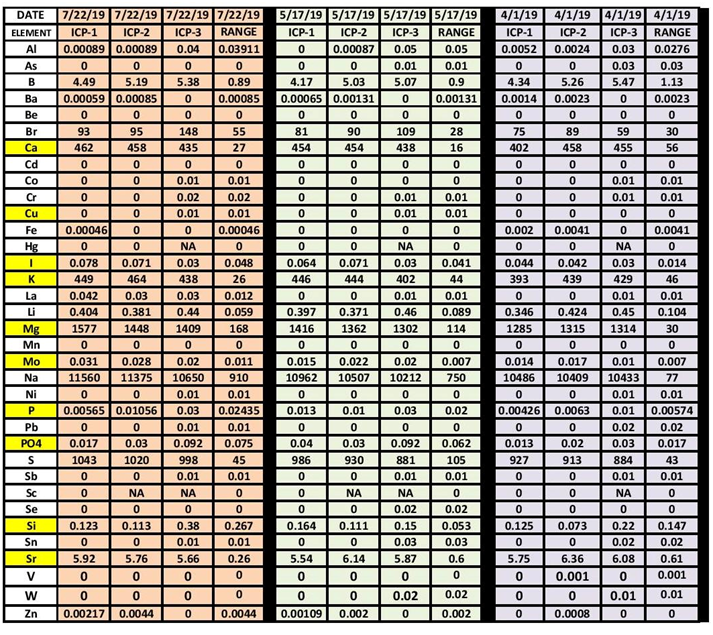
I've seen at least one thread where someone split a sample and sent it to several ICP companies. The magnesium was wildly and disappointingly different.

Here is the post you are taking about
Ok, so you are moving away from their ecosystem. You do not have any personal experience with the trident as you say, you don't own one. Yet you are perpetuating 2nd hand information.
Look - I hear, and actually agree, on some hobbyist over complicating their systems. But it is hobby and people take it to different levels. Same applies to other hobbies be it scuba diving, cars, or stamp collecting. You can have a simple tank and still own a controller or automated testing system.
I believe you should use what works for you. Sounds like you may have found it. We simply disagree on the Trident's accuracy. And yes, this is based on my first hand experience and a few fellow hobbyists. So I'm not all knowing or the Oracle
I do not understand that you post this again - in other threads it has been showed that this is only valid to PO4 reading - not to the total P readings that an ICP test do.Take a look at this post...https://www.reef2reef.com/threads/sample-storage-and-its-impact-on-phosphate-measurement.696800/ This is some pretty strong indication that as @Dan_P said that sample storage (shipment) does have an impact on the measurement results and the samples we send of keep for our own testing are not "stable" for long periods of time.... More work has been done on this question that I believe will clarify the point but it is not yet ready for posting...
Respectfully
rick
I definitely like the idea of making, or buying, standards to check our home test kits.
@Randy Holmes-Farley has shown us how to make an alkalinity standard and I have purchased 3 commercially-made standards: Hanna P, Hanna Alk, and GHL Alk. HOWEVER, these 3 standards all have a "margin of error" of up to 10% which seems to almost make them of less value than I would like.
I have spent a lot of time comparing test kit brands and have found the most variation with nitrate test kits (Nyos, Red Sea and Salifert) which has been very frustrating. When Nyos reads 5, Red Sea will read 8-12 and Salifert will be even higher.
1) Can anyone tell us how to make an accurate NO3 standard, or where one can be purchased (either with an accuracy better than +/- 10%)?
2) And, also for PO4?
Thank you!
Is this standards in 35 psu saltwater? if not - they are IMO worthless because lack of disturbing ions. IMO - the only way to check the accuracy of any test (and especially NO3 tests) is to use your own saltwater and spike the sample with a known amount of the compound you want to test. Its known as the the standard additions method.HACH sells both of these...
rick
This may be a lot to ask someone to check my math.....but if anyone cares to do so, are my calculations for a NITRATE standard (to raise a sample of tank water) correct?:Is this standards in 35 psu saltwater? if not - they are IMO worthless because lack of disturbing ions. IMO - the only way to check the accuracy of any test (and especially NO3 tests) is to use your own saltwater and spike the sample with a known amount of the compound you want to test. Its known as the the standard additions method.
Take first a test of your saltwater - spike it with a known amount - test again. From the difference you can read the accuracy of your tests in your aquarium. As many has stated here - saltwater is a complex matrix of different compounds and these different compounds can interact with the readings of any single compounds with all types if measurements methods - even ICP. It is necessary to know this when you compare different tests and test methods.
Sincerely Lasse
This may be a lot to ask someone to check my math.....but if anyone cares to do so, are my calculations for a NITRATE standard (to raise a sample of tank water) correct?:
Stock Solution: 19.2 g NaNO2 in 500 ml RO/DI
Adding 1 drop of Stock Solution to 10 ml of tank water will raise NO3 by 4 ppm.
Or, if someone has another "already calculated" stock solution, that would be helpful.
Thanks for helping!
If I get it right - at step 3 you have 4.0 gram/L -> 1 ml of this is 4 mg NO3. If you take this (1 ml = 4mg NO3) and fill up to 1000 ml (1L) with aquarium water - you have spike this saltwater with exactly 4 ppmWorking backwards ... you want a 10mL solution at 4ppm NO3 yes?.
1. Start with 4ppm NO3. 4ppm NO3 = 4mg/L NO3 = .004 g/L NO3 = 0.0052 g/L NaNO3
2. Let's do this by serial dilution. Let's create a stock 5.2g/L solution and dilute from there.
3. Dissolve 5.2g NaNO3 in Liter exactly. This 1-L solution is 5.2g/L NaNO3 or 4.0g/L NO3.
4. You want a 4.0 mg/L NO3 .. not 4.0 g/L so take that stock you made in step 3 and dilute by 1,000.
5. 1 mL of stock (4.0g/L NO3) in 1 Liter of rodi is now 4.0 ppm NO3 or 5.2 ppm NaNO3.
Disclaimer... Use this information at your own risk. FYI.. avoid using drop size when possible. a graduated cylinder or syringe is a much better option. Make dilutions with volumes that are easy to measure in a syringe or cylinder>
