- Joined
- May 22, 2016
- Messages
- 6,602
- Reaction score
- 10,193
This topic is @Dan_P 's baby - I'm just running with it. Dan will even go further than I will and use some of the API kits that I won't mess with (NO3, PO4).
It's widely claimed that API tests can't be trusted and any values reported by API should be ignored until the hobbyist throws them in a trashcan and a more expensive test kit is purchased.
But when you measure the color response of these test kits to carefully made stock solutions of known concentration - you find that API kits perform exactly as they should in theory - forming color linearly proportional to the concentration.
I wanted to illustrate this by demonstrating the colorimetric performance of Total Ammonia, Nitrite, and High Range pH tests.
Here's API total ammonia test
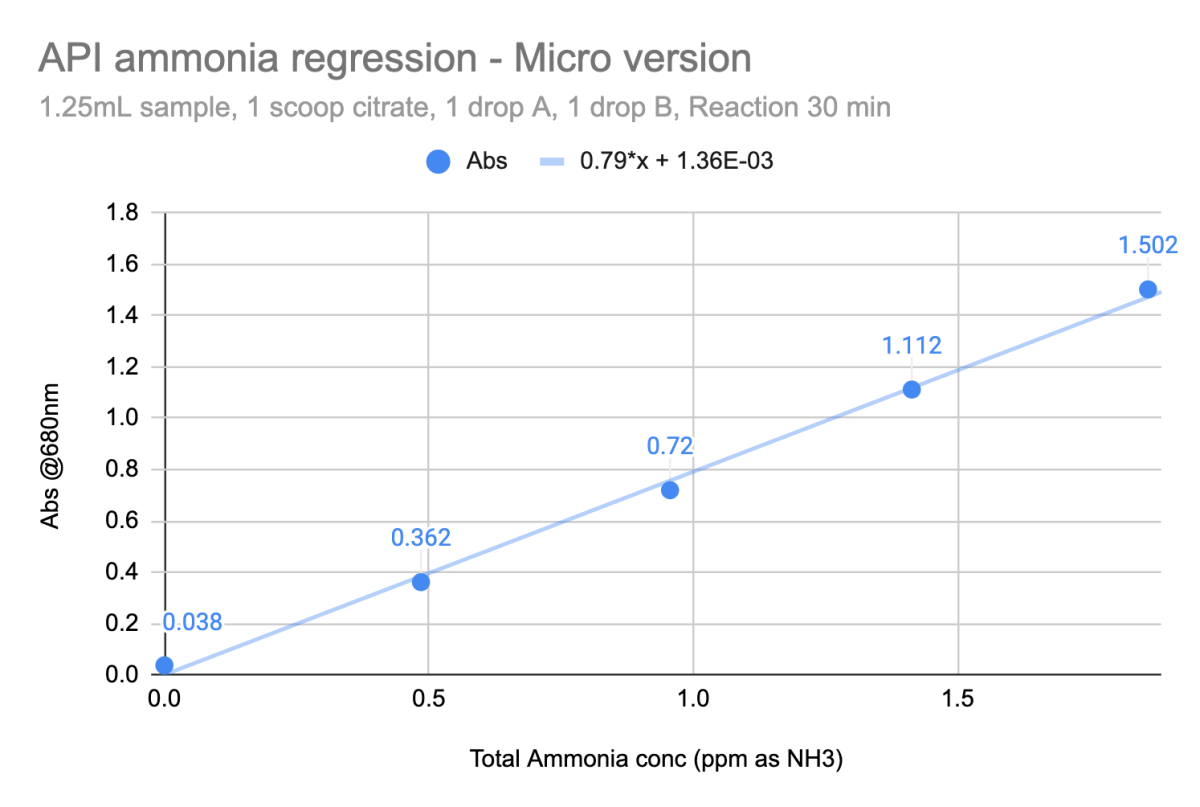
Tightly linear over the range [0 - 2ppm]. The reagent amounts and ratios are tweaked a bit from the API box instructions for the range and the volume that I want to work with, but all the reagents are API. And whatever box of reagents I grab the performance is the same.
Next is the Nitrite - NO2 test.
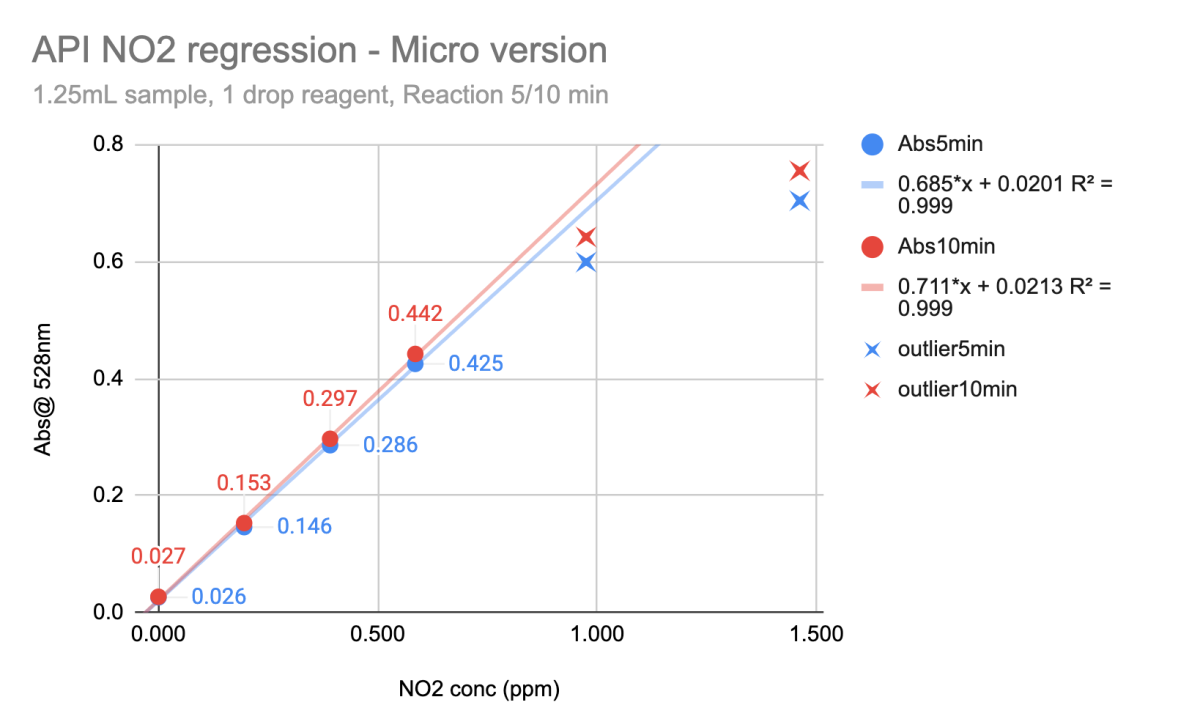
With the recipe I'm working with, it stops being perfectly linear around 1ppm NO2 and above, so I use this when working with samples between [0 - 0.7ppm]. Above 1ppm, it still gets darker pink, just not in a clean linear way (at this reagent ratio).
And finally, here's API high range pH.
The pH test is a color indicator that's phenol red or very similar - it responds very well over the entire plausible range of saltwater pH. Here's the absorbance spectrum of the indicator in saltwater from pH of less than 7.0 to above 8.6.
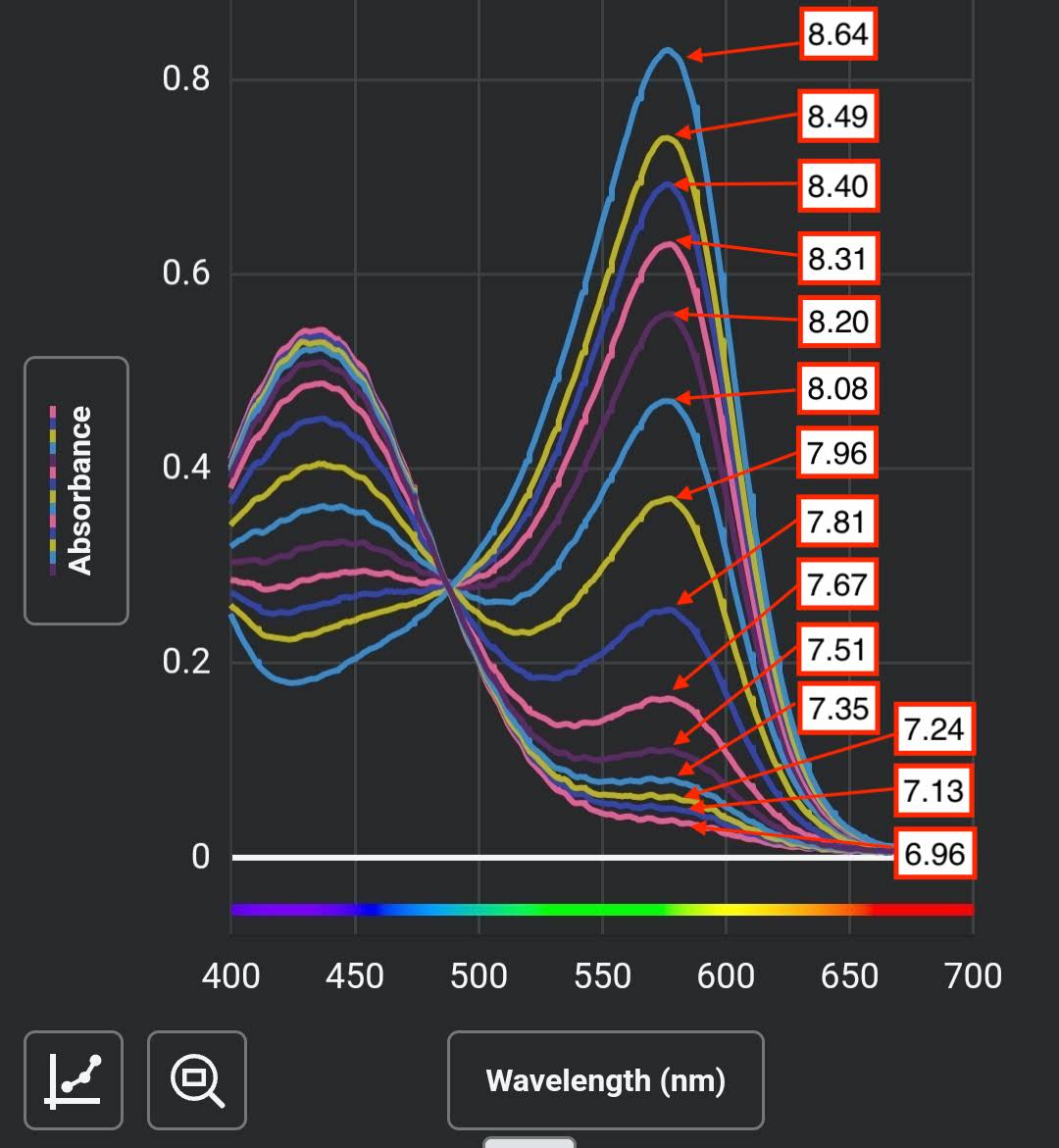
The plot is showing the spectral data, but each of those colors is easily naked-eye distinguishable by comparison as well.
So if you do a ratio of the left peak and right peak absorbance and plot the log of that vs what a calibrated pH meter reads in the same solution you get this...
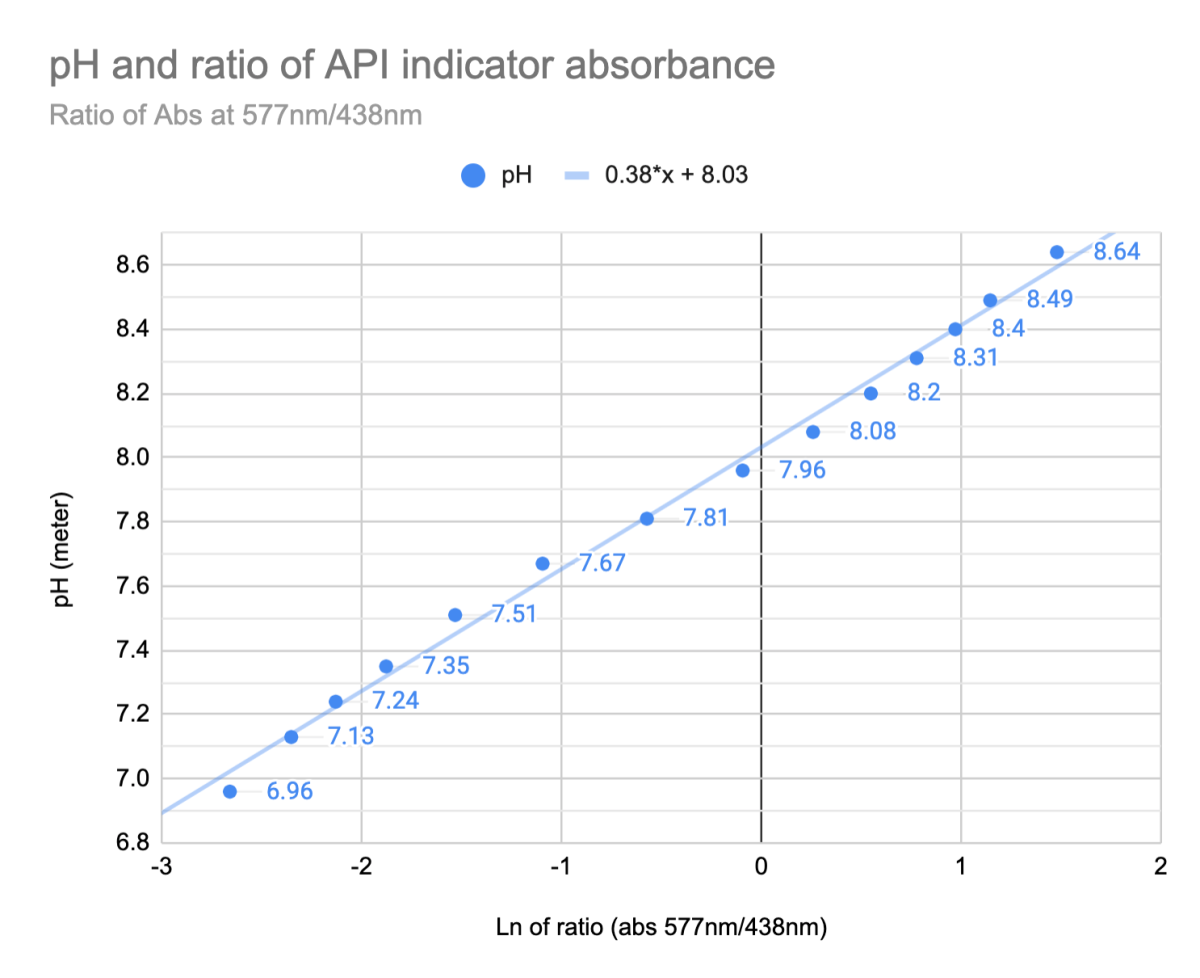
The log of the absorbance ratio is tightly linear to what my calibrated pH meter gives - within 0.05 pH units. (This means pH can be measured by recording color and no probe/calibration solution required, which is convenient sometimes.)
In all these examples, the technical details are unimportant, or at least the topic for another thread. What matters is that in all cases the API reagents are doing exactly what you want a chemical reagent to do. They have a repeatable, easily distinguished color response that is linear to the concentration of what you want to measure.
Here's an absurd example to drive the point home about the gap between how low the trust is of API vs how consistent the performance actually is.
I found this in a box in my garage: From the Lot numbers and the copyright info on the box and printed inserts - it was made in 2003.
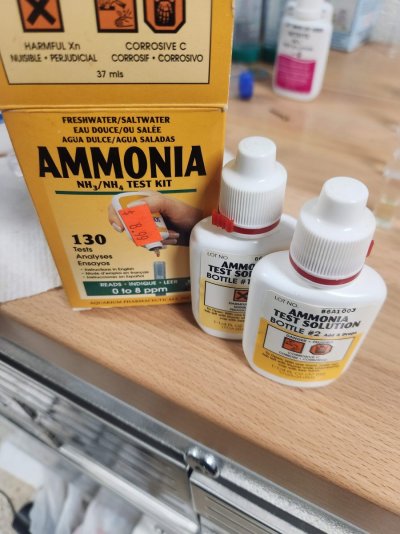
So, how well does this 20yr old API ammonia kit work?
I used both API kits made 20 years apart to measure the same 0.0 and 1.0 ppm total ammonia saltwater solutions.

Pic taken at around 10 minutes - colorimetric measurements were made at around 30 minutes and I was a little shocked to see both kits gave completely identical results. ( I expected that the chlorine solution in reagent 2 would have lost its potency, but I guess the bottle was well sealed ... for 20 years. lol)
So when we say API results are "trash" etc (and there are some nonsensical results from API tests posted), we should probably talk about what we mean by that, and why they might be bad - because the chemistry is solid.
So telling somebody to go use a different set of chemicals to do the same thing seems unlikely to give a better result if the chemicals weren't the problem in the first place, right?
Dr. Wellfish says "Your garbage test results aren't my fault."
[Disclaimer, I don't love all API kits: NO3 and PO4 have too much lot to lot variation - so I'll always opt for hanna / red sea there.]
It's widely claimed that API tests can't be trusted and any values reported by API should be ignored until the hobbyist throws them in a trashcan and a more expensive test kit is purchased.
But when you measure the color response of these test kits to carefully made stock solutions of known concentration - you find that API kits perform exactly as they should in theory - forming color linearly proportional to the concentration.
I wanted to illustrate this by demonstrating the colorimetric performance of Total Ammonia, Nitrite, and High Range pH tests.
Here's API total ammonia test
Tightly linear over the range [0 - 2ppm]. The reagent amounts and ratios are tweaked a bit from the API box instructions for the range and the volume that I want to work with, but all the reagents are API. And whatever box of reagents I grab the performance is the same.
Next is the Nitrite - NO2 test.
With the recipe I'm working with, it stops being perfectly linear around 1ppm NO2 and above, so I use this when working with samples between [0 - 0.7ppm]. Above 1ppm, it still gets darker pink, just not in a clean linear way (at this reagent ratio).
And finally, here's API high range pH.
The pH test is a color indicator that's phenol red or very similar - it responds very well over the entire plausible range of saltwater pH. Here's the absorbance spectrum of the indicator in saltwater from pH of less than 7.0 to above 8.6.
The plot is showing the spectral data, but each of those colors is easily naked-eye distinguishable by comparison as well.
So if you do a ratio of the left peak and right peak absorbance and plot the log of that vs what a calibrated pH meter reads in the same solution you get this...
The log of the absorbance ratio is tightly linear to what my calibrated pH meter gives - within 0.05 pH units. (This means pH can be measured by recording color and no probe/calibration solution required, which is convenient sometimes.)
In all these examples, the technical details are unimportant, or at least the topic for another thread. What matters is that in all cases the API reagents are doing exactly what you want a chemical reagent to do. They have a repeatable, easily distinguished color response that is linear to the concentration of what you want to measure.
Here's an absurd example to drive the point home about the gap between how low the trust is of API vs how consistent the performance actually is.
I found this in a box in my garage: From the Lot numbers and the copyright info on the box and printed inserts - it was made in 2003.

So, how well does this 20yr old API ammonia kit work?
I used both API kits made 20 years apart to measure the same 0.0 and 1.0 ppm total ammonia saltwater solutions.
Pic taken at around 10 minutes - colorimetric measurements were made at around 30 minutes and I was a little shocked to see both kits gave completely identical results. ( I expected that the chlorine solution in reagent 2 would have lost its potency, but I guess the bottle was well sealed ... for 20 years. lol)
So when we say API results are "trash" etc (and there are some nonsensical results from API tests posted), we should probably talk about what we mean by that, and why they might be bad - because the chemistry is solid.
So telling somebody to go use a different set of chemicals to do the same thing seems unlikely to give a better result if the chemicals weren't the problem in the first place, right?
Dr. Wellfish says "Your garbage test results aren't my fault."

[Disclaimer, I don't love all API kits: NO3 and PO4 have too much lot to lot variation - so I'll always opt for hanna / red sea there.]
Last edited:


















