Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
GHL KHD
- Thread starter Aaronreef
- Start date
- Tagged users None
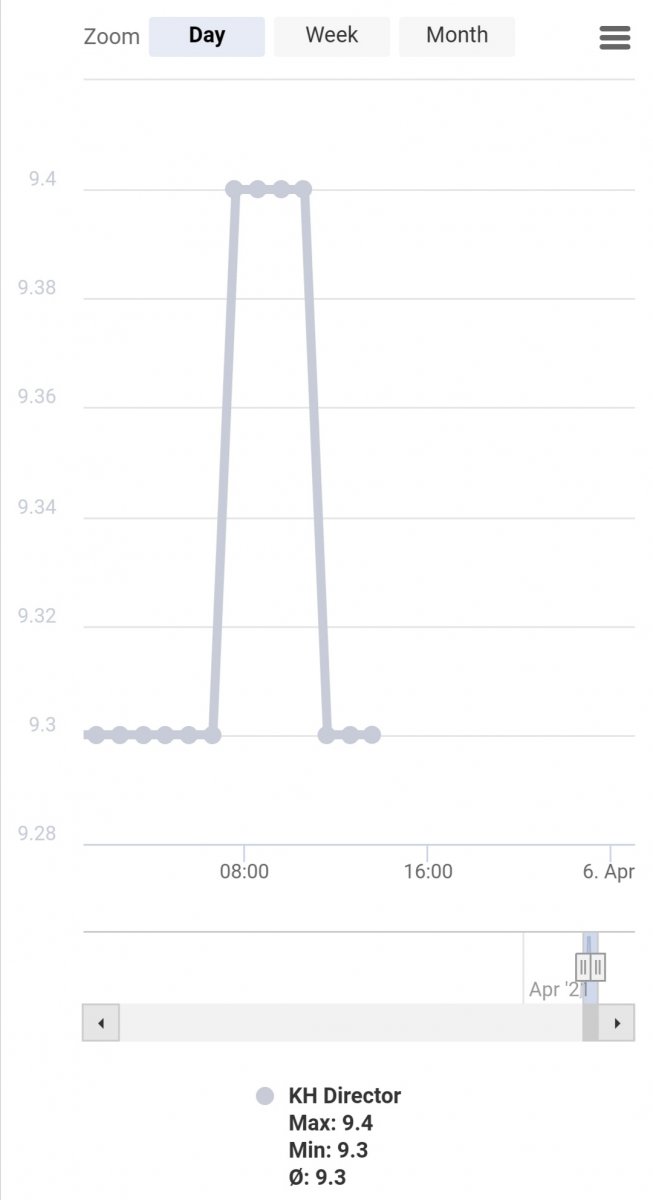
Mines set to 100ml,sample line is in a low flow part of sumpi have mine on a angle just below the water it’s held in place with a suction cupIs the amount of sample water I'm using ok 100ml
- Joined
- Apr 16, 2019
- Messages
- 351
- Reaction score
- 102
Just started again testing the tank will leave it over night and see what the outcome is.
Another question.
When the sample water is draw into the unit should the 'current ph-value' shown in the khd be the same as my tank pH
Another question.
When the sample water is draw into the unit should the 'current ph-value' shown in the khd be the same as my tank pH
Honestly not sure mate have you tested the ph calibration fluid to say that’s what the probe is readingJust started again testing the tank will leave it over night and see what the outcome is.
Another question.
When the sample water is draw into the unit should the 'current ph-value' shown in the khd be the same as my tank pH
- Joined
- Apr 16, 2019
- Messages
- 351
- Reaction score
- 102
Yes recalibrated the probe again today.
The 'current pH value' reads as it should when in a calibration fluid.
First test complete on the tank and is giving a read of what is expected inline with the Hanna checker
Will leave it over night to test and see the outcome tomorrow
Really appreciate all your help by the way
The 'current pH value' reads as it should when in a calibration fluid.
First test complete on the tank and is giving a read of what is expected inline with the Hanna checker
Will leave it over night to test and see the outcome tomorrow
Really appreciate all your help by the way
I would just leave it now for a couple of days,every time I adjust mine takes a few tests to settle again,when you got the low reading it could be possible it sucked up a air bubble
- Joined
- Apr 16, 2019
- Messages
- 351
- Reaction score
- 102
I'll leave as Is for now.
If it does seem to be better then maybe it was air.
If that's the case what's the longest I can go with the water sample line?
I have to have the unit above the tank as I haven't got any space anywhere else
If it does seem to be better then maybe it was air.
If that's the case what's the longest I can go with the water sample line?
I have to have the unit above the tank as I haven't got any space anywhere else
Honestly not sure on that maybe some one else could say,I would say it would be fine just recalibrate pumps and weight every thing againI'll leave as Is for now.
If it does seem to be better then maybe it was air.
If that's the case what's the longest I can go with the water sample line?
I have to have the unit above the tank as I haven't got any space anywhere else
Yea fingers crossed I find any adjustment on them they need a day to settleSo far so good since yesterday, of it stay like this I'd be happy.
Thanks again @paul01609 for all the help
I've got an answer from him on Youtube. I think he misunderstood one detail. CO2 will drop pH, yes, it will form some carbonic acid. But even though it's an acid, it will not drop your alkalinity. H2CO3 -> H + HCO3 so there's an equilibrium. for every H+ that comes out of carbonic acid and reacts with bicarbonate, you get another carbonic acid and another bicarbonate. you just switch H from one molecule to another. No change in alkalinity, but pH does drop.Not all alk drops are caused by coral consumption. Low pH apparently binds carbonates to the rock. To what extent? I don't know.
I remember seeing Chris Meckley saying this and it seems counterintuitive. After all shouldn't low pH dissolve coral skeletons?
But he was saying that thing binding happens at high 7's.
He was saying that when you manage to raise the pH of your tank to 8.3 and above, the carbonates that were bound to the rock and sand are released.
Similar threads
- Replies
- 4
- Views
- 280
- Price: 1,000
- Shipping Available
- Replies
- 2
- Views
- 171
- Price: 550
- Shipping Available
- Replies
- 0
- Views
- 94
California Aquarium Controller Filters Testing Package Deal Drygoods
FS Clarisea SK, alkatronic GHL hydros, priced lowered!
- Price: $200+
- Shipping Available
- Replies
- 6
- Views
- 1,160

















