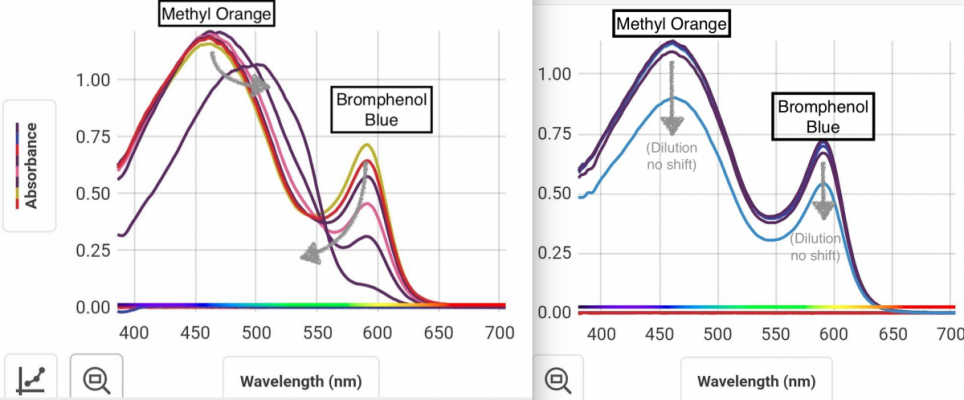
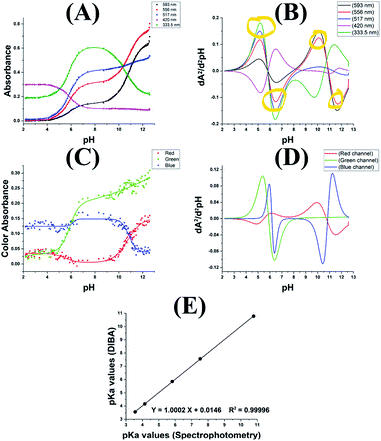
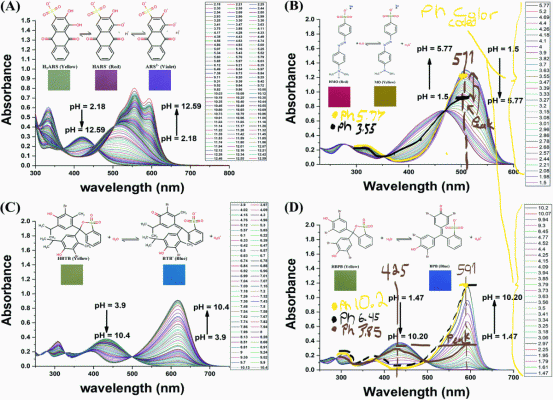
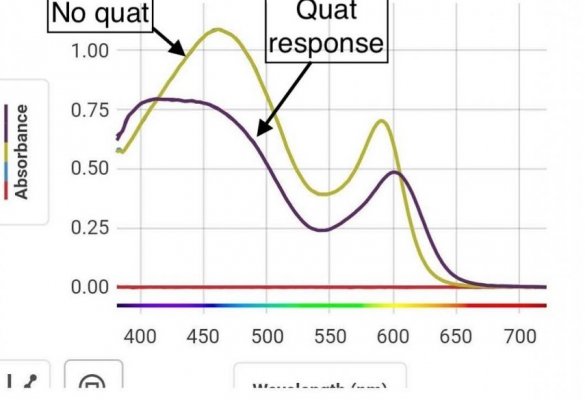
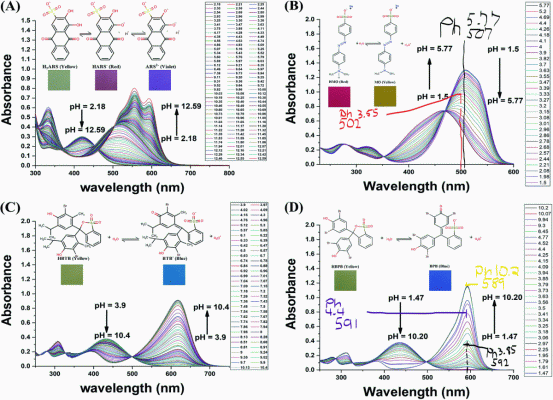
Mixture of two indicators, again think color wheel. Your other vials don't look far off from picture I linked with just MO vs ph. If you hit this just right it will show green, and can go back. This is not a reaction with the indicators. It is merely a charged state of the indicator bending the molecule and changing the energy. This is why we see color change in this case.green
Things that usually have a hydrogen are acids, or a compound that is more electronegative. Its actually complicated. Like when you say way way earlier bpb " it is changing to its acid form" It was likely already acidic, but the aspartic acid was changing charges. Polyquaternium-42 or 60 is basic. "Basic form indicators" like you say would not change colors if what you said was true. They would be the color of their neutral forms, or the color of positive/negative charge form. PH dependent of course.MO and BPB are in the basic forms
1: I only ever said I think these are not in there.
2: Why do you avoid answering some questions?
3: I am only pointing out what you have done is not a conclusion of anything, but you are so stuck to it I don't know what to tell you.
4: Thank you @UWC I am not trying to defend you, I think you can manage. It seems you are confident it is not in there.