Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,430
- Reaction score
- 63,792
Reef Chemistry Question of the Day [HASHTAG]#172[/HASHTAG]
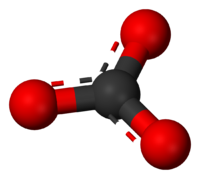
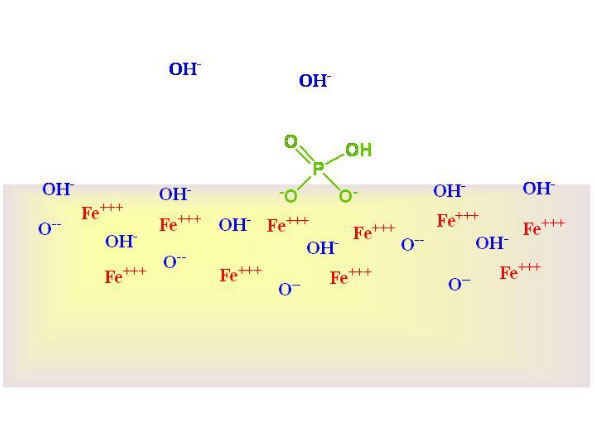
Phosphate is known to bind to the surface of calcium carbonate, and this binding to rock and sand can have significant implications for the phosphate cycle in reef aquaria.
Which arrangement of atoms is most likely to be holding phosphate to the calcium carbonate surface?
A. Calcium binding to phosphorus binding to oxygen
B. Calcium binding to oxygen binding to phosphorus
C. Carbon binding to phosphorus binding to oxygen
D. Carbon binding to oxygen binding to phosphorus
E. Oxygen binding to phosphorus binding to calcium
F. Oxygen binding to phosphorus binding to carbon
Good luck!
.
Phosphate is known to bind to the surface of calcium carbonate, and this binding to rock and sand can have significant implications for the phosphate cycle in reef aquaria.
Which arrangement of atoms is most likely to be holding phosphate to the calcium carbonate surface?
A. Calcium binding to phosphorus binding to oxygen
B. Calcium binding to oxygen binding to phosphorus
C. Carbon binding to phosphorus binding to oxygen
D. Carbon binding to oxygen binding to phosphorus
E. Oxygen binding to phosphorus binding to calcium
F. Oxygen binding to phosphorus binding to carbon
Good luck!
.
Last edited: