- Joined
- May 22, 2016
- Messages
- 6,545
- Reaction score
- 10,101
In normal Tank water, ORP is slow to equalize and is affected by so many things that the number itself tells you very little of interest.
But if you add a strong oxidizing agent to a sample of tank water, then the ORP can be really informative - it can tell you how that oxidizer is reacting with substances in the sample. Chlorine (bleach) and its reactions are well-studied in water treatment and it reacts quickly with several things that show up in normal tank water and could be worth knowing.
(I'll type up a procedure in a follow-up post for anyone else who wants to try it. I'm curious what other people's tank water looks like.
edit: procedure detailed in post 11)
Let's look at how ORP behaves with chlorinating a few different samples. Each data point is an addition of bleach solution done every 30 seconds. The available chlorine or free chlorine added is noted as Cl2 (ppm)
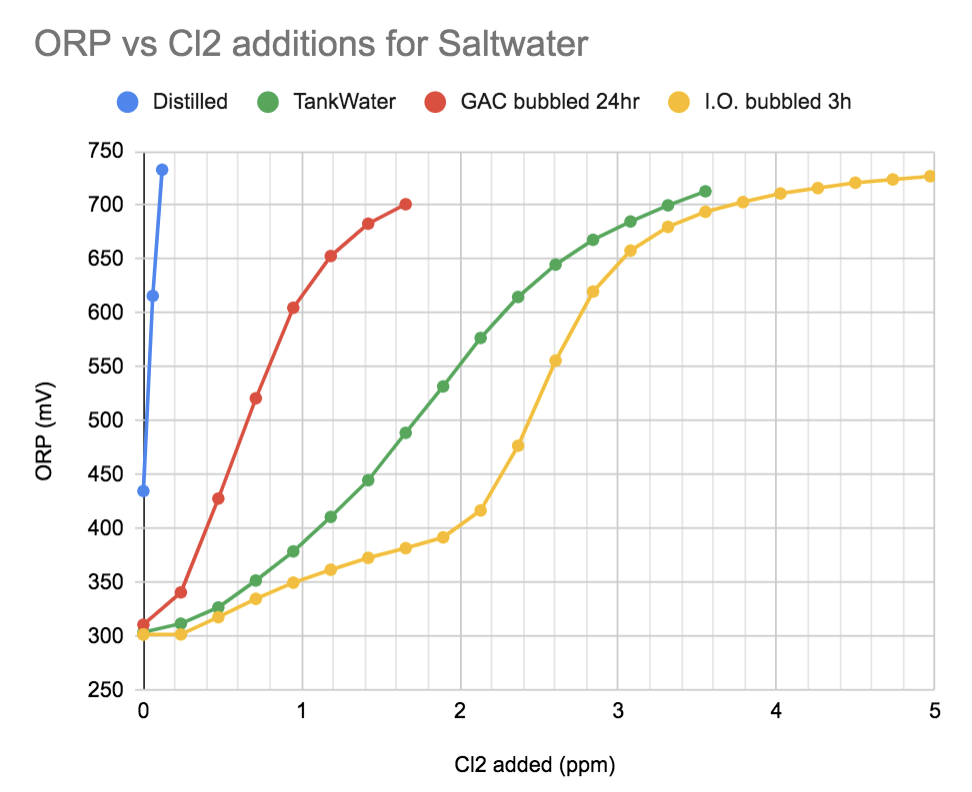
It takes barely any Cl2 to raise the ORP of distilled water (Blue)
My tank water (Green)
1L of that tank water placed in a beaker with 10mL GAC and bubbled for 24 hours greatly reduced the amount of Cl2 reactive compounds in my tank water (Red line)
Fresh Instant Ocean at matching 1.026sg to my tank water, bubbled for 3 hours contains an interestingly large amount of Cl2 reacting compounds. (Yellow)
So what sorts of things is the Cl2 reacting with? Since I.O. is complicated, I'll use the GAC-treated tank water as a baseline.
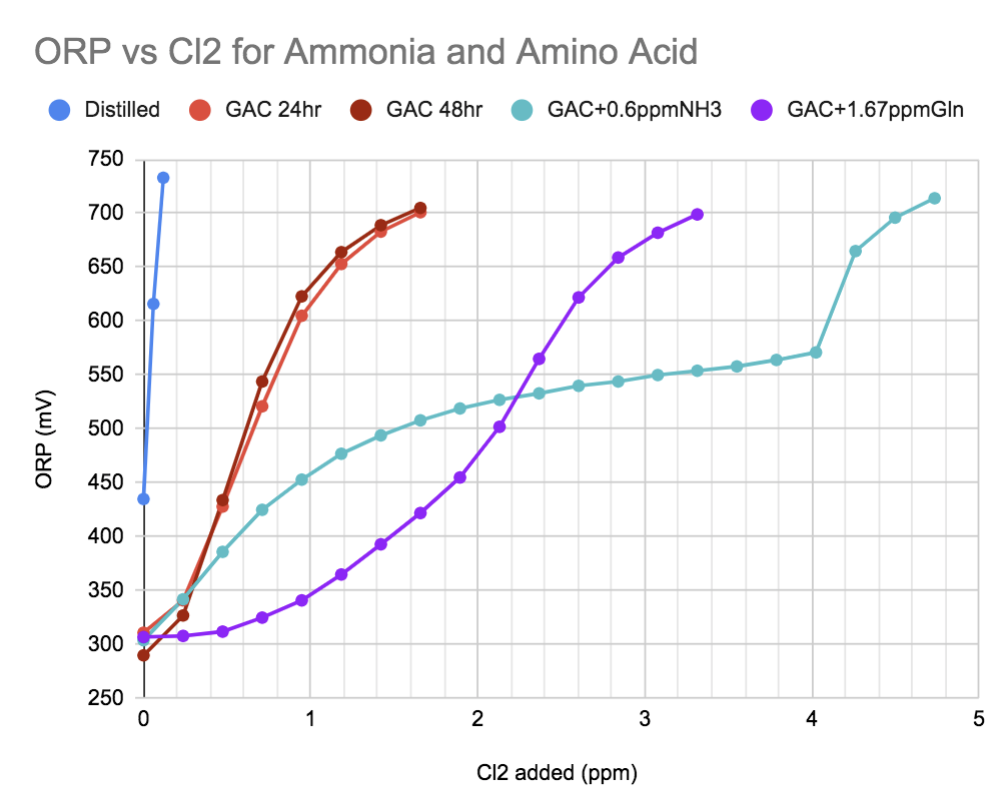
In Red are the GAC-bubbled tank water samples at 24 and 48hrs. Very little additional removal of organics occurred from 24-48 hr.
In Teal, the GAC baseline water spiked with 0.6ppm total ammonia (calculated as NH3ppm) has this really eye-catching shift at 600-650mV.
In Purple is the baseline water spiked with 1.67ppm of the amino acid Glutamine.
As you can see, the chemistry of chlorination is really sensitive to these N-containing compounds, and the details of the reaction show up in the ORP. The rapid jump in the ORP of the chlorinated ammonia sample apparently correlates to the destruction of chloramine (google image from a paper).
In fact the amount of additional Cl2 required to raise the ORP of ammonia and amino acids seems to agree with chemistry theory fairly well.
If you convert concentrations in the above graph to moles, you get in the ballpark of 1.28 Cl2 per ammonia (theory is 1.5) and about 2.04 Cl2 per glutamine (theory is 2 - in short time).
What else does and doesn't react with Chlorine that might matter for tank water.
The major nutrients PO4 and NO3 seem totally unreactive with Cl2 over a short period.
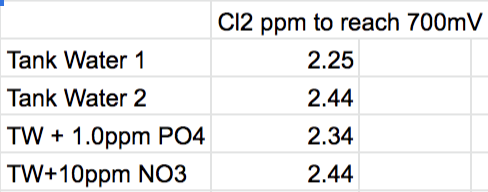
Adding PO4 of 1ppm and NO3 of 10ppm didn't have any effect on the amount of Cl2 required to raise ORP of tank water (within uncertainty).
What does affect it?
Ammonia and N-containing organics like amino acids and proteins. I don't have details on proteins but I can say that it's sensitive to what accumulates in skimmate, what is removed by GAC and what's added to the water 1hr after feeding fish flake (but not NO3 & PO4).
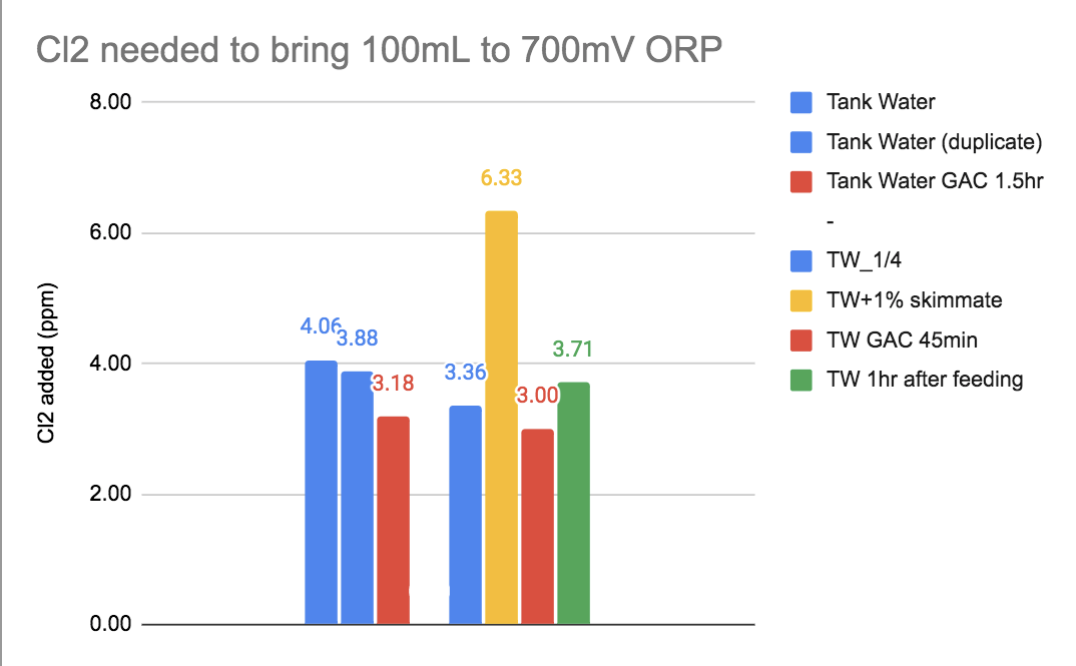
(note: after seeing more data, I would now probably pick a lower target than 700mV for this measure - too flat at 700mV, but the above data illustrates the point regardless.)
I'll wrap up for now. Anyone else tried similar stuff?
(credit to @Dan_P for prodding me into looking at Cl2 consumptions of aquarium water)
But if you add a strong oxidizing agent to a sample of tank water, then the ORP can be really informative - it can tell you how that oxidizer is reacting with substances in the sample. Chlorine (bleach) and its reactions are well-studied in water treatment and it reacts quickly with several things that show up in normal tank water and could be worth knowing.
(I'll type up a procedure in a follow-up post for anyone else who wants to try it. I'm curious what other people's tank water looks like.
edit: procedure detailed in post 11)
Let's look at how ORP behaves with chlorinating a few different samples. Each data point is an addition of bleach solution done every 30 seconds. The available chlorine or free chlorine added is noted as Cl2 (ppm)
It takes barely any Cl2 to raise the ORP of distilled water (Blue)
My tank water (Green)
1L of that tank water placed in a beaker with 10mL GAC and bubbled for 24 hours greatly reduced the amount of Cl2 reactive compounds in my tank water (Red line)
Fresh Instant Ocean at matching 1.026sg to my tank water, bubbled for 3 hours contains an interestingly large amount of Cl2 reacting compounds. (Yellow)
So what sorts of things is the Cl2 reacting with? Since I.O. is complicated, I'll use the GAC-treated tank water as a baseline.
In Red are the GAC-bubbled tank water samples at 24 and 48hrs. Very little additional removal of organics occurred from 24-48 hr.
In Teal, the GAC baseline water spiked with 0.6ppm total ammonia (calculated as NH3ppm) has this really eye-catching shift at 600-650mV.
In Purple is the baseline water spiked with 1.67ppm of the amino acid Glutamine.
As you can see, the chemistry of chlorination is really sensitive to these N-containing compounds, and the details of the reaction show up in the ORP. The rapid jump in the ORP of the chlorinated ammonia sample apparently correlates to the destruction of chloramine (google image from a paper).
In fact the amount of additional Cl2 required to raise the ORP of ammonia and amino acids seems to agree with chemistry theory fairly well.
If you convert concentrations in the above graph to moles, you get in the ballpark of 1.28 Cl2 per ammonia (theory is 1.5) and about 2.04 Cl2 per glutamine (theory is 2 - in short time).
What else does and doesn't react with Chlorine that might matter for tank water.
The major nutrients PO4 and NO3 seem totally unreactive with Cl2 over a short period.
Adding PO4 of 1ppm and NO3 of 10ppm didn't have any effect on the amount of Cl2 required to raise ORP of tank water (within uncertainty).
What does affect it?
Ammonia and N-containing organics like amino acids and proteins. I don't have details on proteins but I can say that it's sensitive to what accumulates in skimmate, what is removed by GAC and what's added to the water 1hr after feeding fish flake (but not NO3 & PO4).
(note: after seeing more data, I would now probably pick a lower target than 700mV for this measure - too flat at 700mV, but the above data illustrates the point regardless.)
I'll wrap up for now. Anyone else tried similar stuff?
(credit to @Dan_P for prodding me into looking at Cl2 consumptions of aquarium water)
Last edited:

















