After our conversation last week I did some reading and am hoping for a little clarification on why you made the above statement. I read your excellent article on alkalinity. https://reefs.com/magazine/chemistry-and-the-aquarium-what-is-alkalinity/I don’t think consumption of alkalinity has much if anything to do with why we get pH swings. It’s variable amounts of CO2.
In the article you provide a graph that shows you adding acid to a saltwater sample and an un-buffered pure freshwater sample. The pH declines gradually in the saltwater sample but drops precipitously in the freshwater sample.
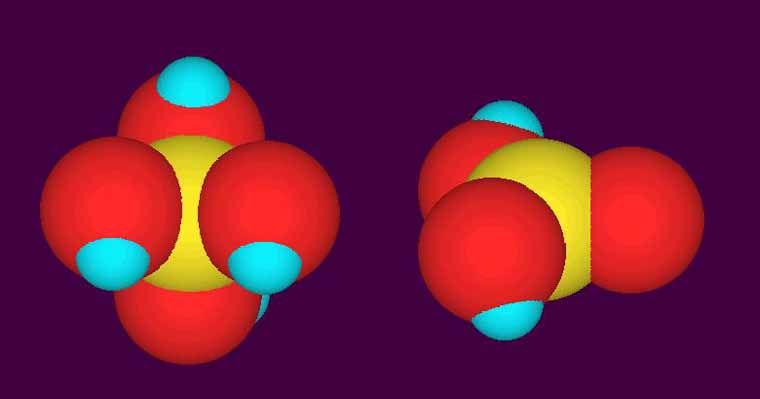
My line of reasoning is that corals remove alkalinity from the water column and incorporate it as calcium carbonate into their skeletons. As the alkalinity ( mostly carbonates and bicarbonates as I understand) is removed from the water, the pH buffering capacity of the water is reduced. Therefore the water is subject to more violent pH swings with the introduction of carbon dioxide from the respiration of organisms and other biological processes.
I guess a more specific question is that if you add a set amount of CO2 to 7dkh water and the same amount to 10dkh water, will there be a more significant pH change in the less buffered water?
Thank you in advance for you time.