Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,600
- Reaction score
- 64,065
Can you link me to a study showing this, or explaining it? So far i have not found any data to show this occurring.
OK, I'll try explaining again. You linked an earlier thread of mine where I discussed how acetate dosing lowers pH and why. That's a fine place to start. Formate is a smaller effect, but in the same direction.
The equation I posted above shows that from a pH perspective, formate dosing for alkalinity it is exactly the same as dosing bicarbonate.
I show in this article below how different alkalinity additives (bicarbonate, carbonate, and hydroxide) impact pH, and this is the bicarbonate section:
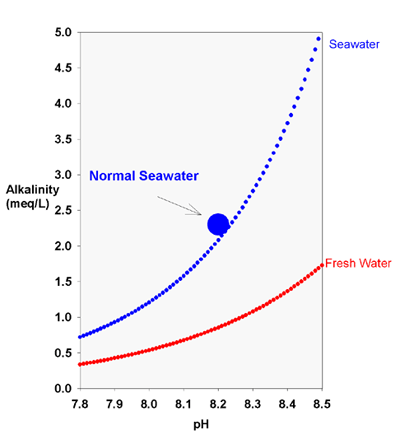
Chemistry And The Aquarium: The Relationship Between Alkalinity And pH
Randy answers many questions in this complex relationship.
Bicarbonate Addition
The addition of bicarbonate as an alkalinity supplement is rather different. In this case, the bicarbonate partially dissociates into carbonate and H+, and the tank experiences an increase in bicarbonate and carbonate, and a drop in pH.:
15. HCO3– → H+ + CO3—
Consequently, the immediate effect on pH is for it to drop. The drop is small because not much of the bicarbonate dissociates at normal tank pH, but enough does to drive the pH a bit lower (from 8.10 to 8.06 in the experiment above).
In the long term, however, the effect is different. Since a substantial amount of bicarbonate was added and the pH did not change much, the tank is now overloaded with bicarbonate with respect to what it would normally have in equilibrium with air. Some of the bicarbonate picks up a proton, becomes carbonic acid, and the pH rises as the CO2 is blown off to the atmosphere:
16. HCO3– + H+ → H2CO3 → CO2 + H2O
In the experiment above, this effect has caused the pH to rise from 8.06 to 8.33. So the long-term effect of bicarbonate addition (as it is for any addition to carbonate alkalinity) is to raise pH even though the short-term effect was to lower it.


















