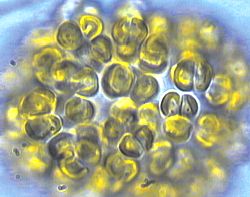
A wikipedia image for chrysophytes.
For some time, I've been dealing with a mystery ailment in my aquarium. A wispy, slimy mass grew on almost every exposed area of my tank - no matter what I had tried, it kept coming back and stubbornly stuck with me for months.
I started my current tank in late May, early June of 2016 from completely dead material - dead rock, no sand, and a small refugium with macro algae. I was running carbon and GFO in a reactor, and had a reef octopus HOB 100 skimmer. Nutrients were almost always undetectable.
Fast forward to August. The macroalgae has been slowly turning white and breaking off, getting into the display tank. I decided to remove it from the refugium section of the sump since it clearly isn't getting enough of what it needs to grow.
Within the week, the chrysophytes moved in.
This was a video I took prior to have it checked out. It had been in the tank for months. Never bothering coral or fish, it covers nearly every free space in the aquarium. Manual removal would help for a day or two, but it'd be back as strong as ever in a few days.
I waited as long as I did thinking it would clear up on its own, but I was fast becoming annoyed and decided to start reaching and to get a positive ID so I could try another treatment.
I reached out to Dr. Capman, a member of our local reefing forum and biology professor at Augsburg College in Minneapolis. He invited me to bring a sample in so we could check it out under a few microscopes.
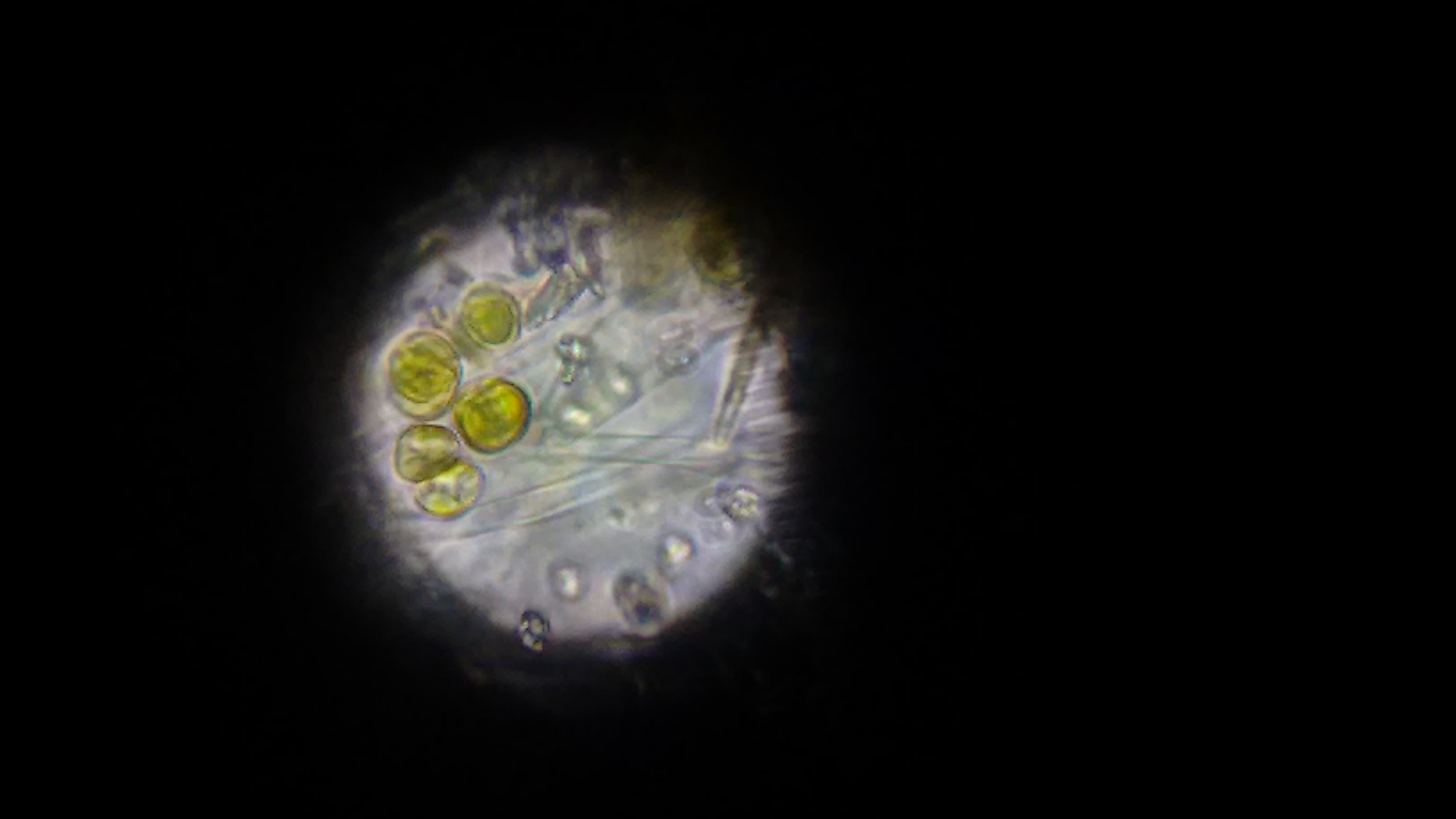
The first image, taken via cellphone through the optics on a microscope.
At first, we didn't know what we were looking for. There were a bunch of these cells, which look awfully similar to the wikipedia image for chrysophytes. At the time, we didn't know what we were looking at. Also in the sample were copious amounts of these long clear filaments. This might be what was giving the golden cells a structure to grow - we weren't sure.
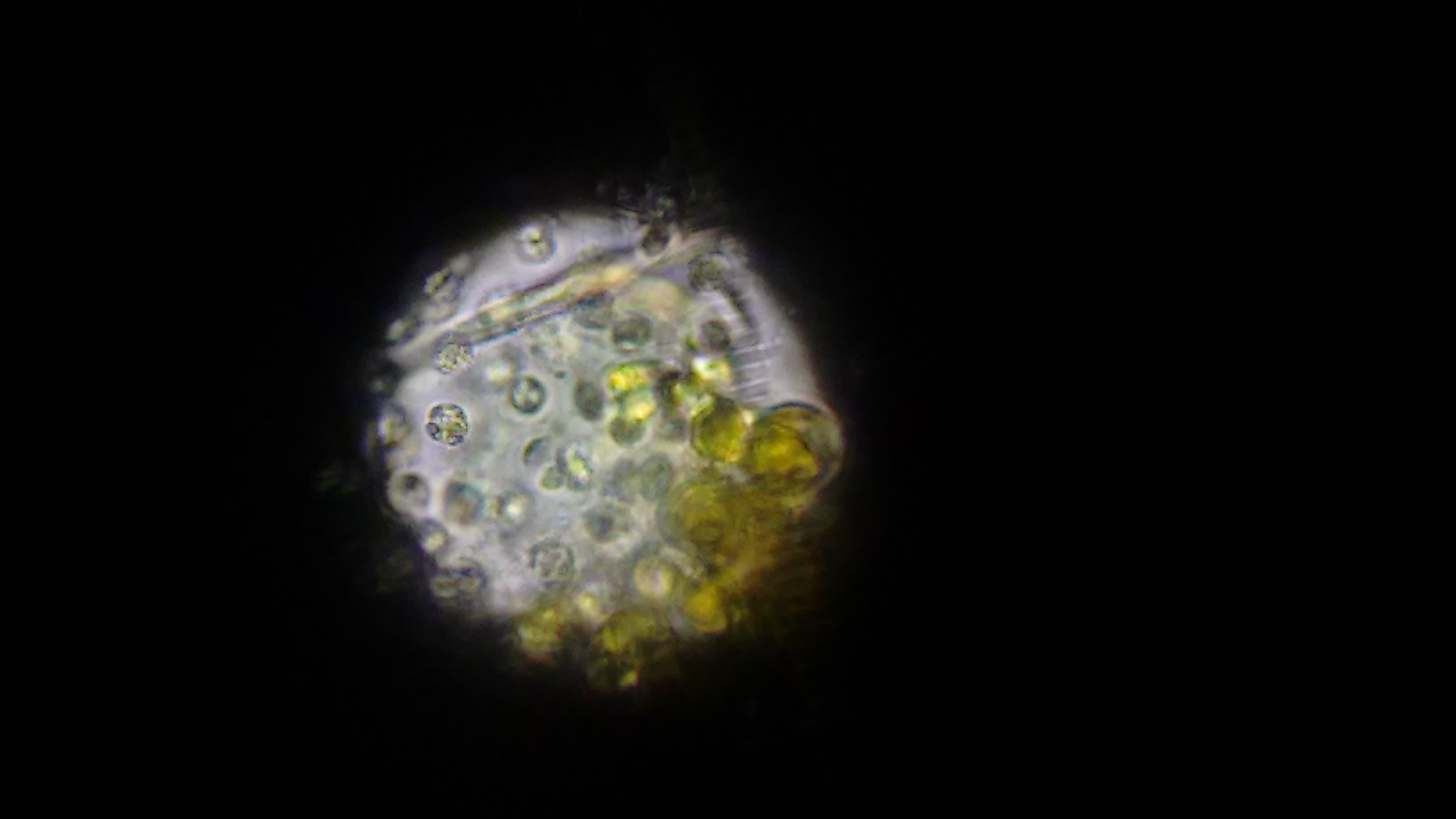
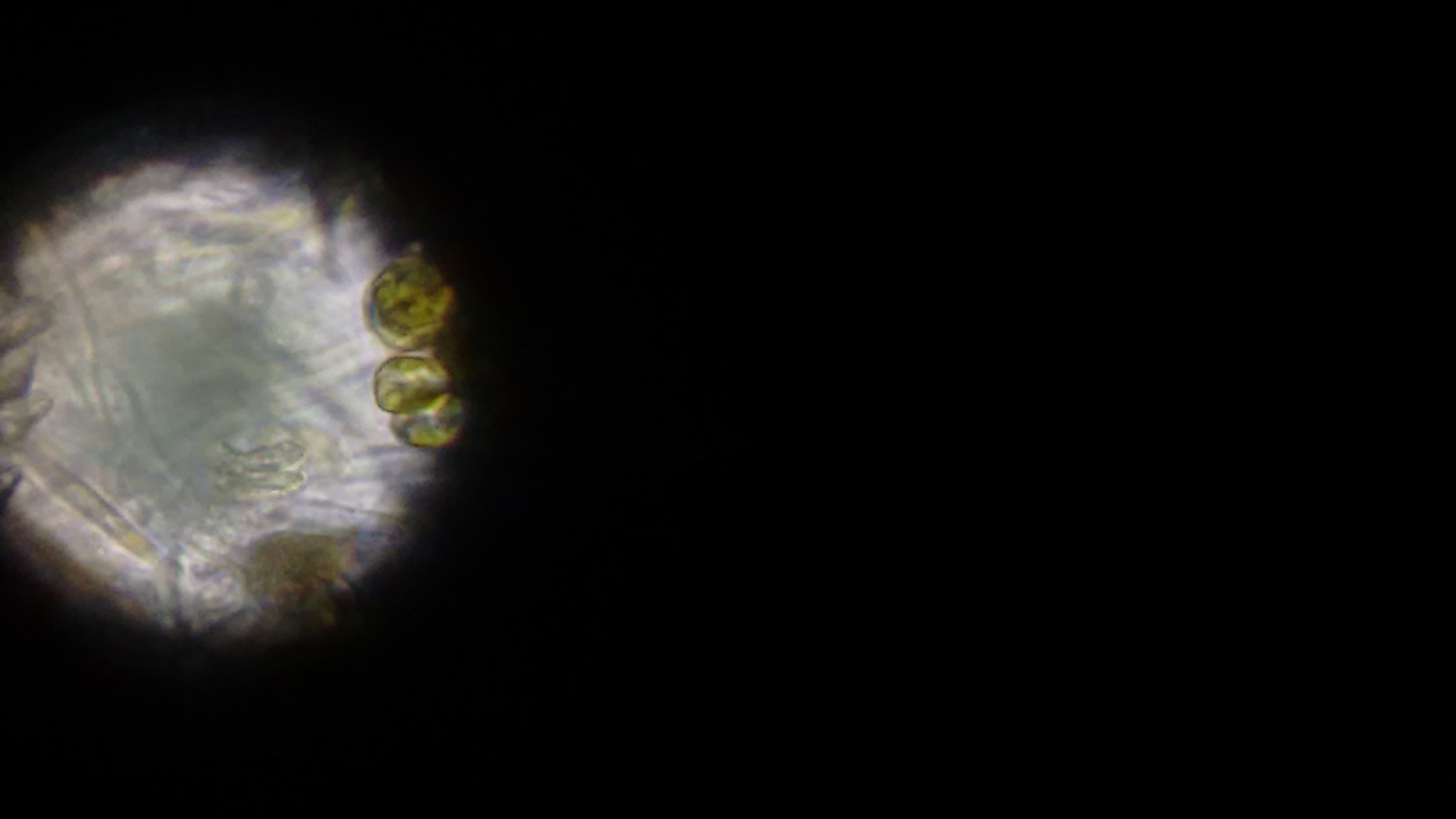
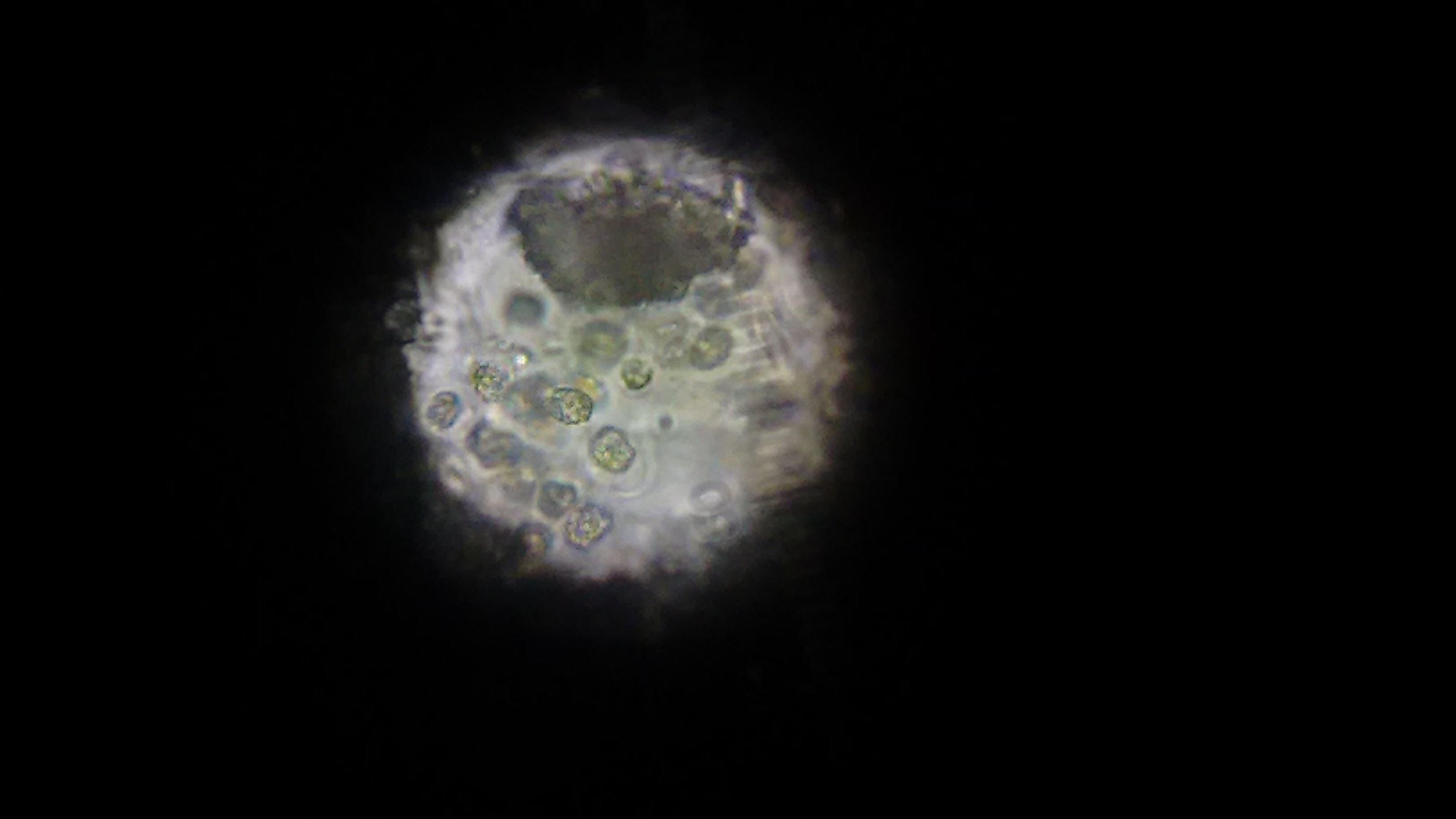
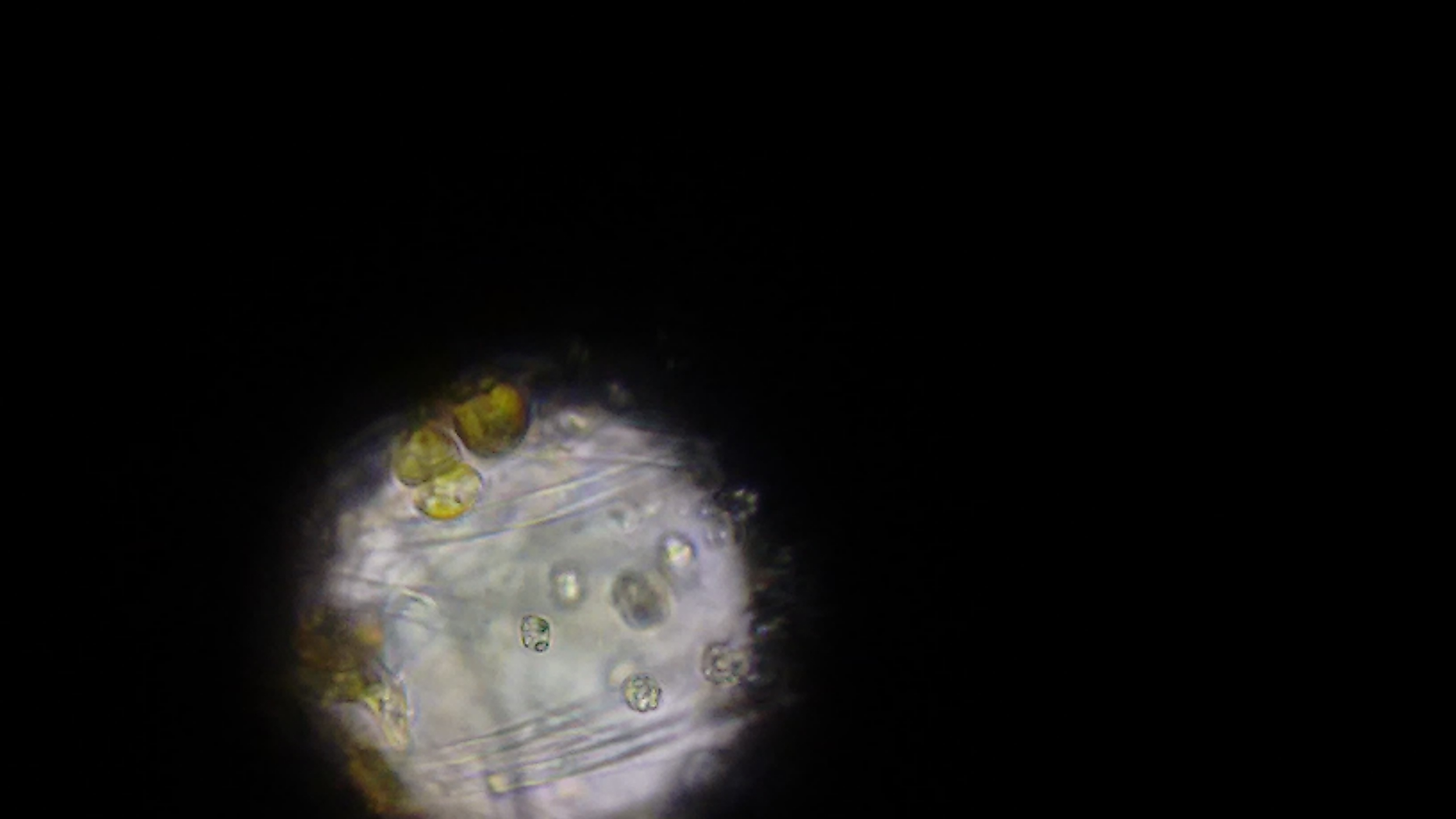

At this point, we ruled out that it was cyano. Not quite sure what it was, we went and used a much more powerful microscope that could send different, specific wavelengths of light through the sample to see what chemicals are excited and fluoresce - potentially telling us what chemicals might be in the cells. (Forgive me, I can't remember the name of the scope, I'm but a simple software developer and not a biologist and terrible with names.

Visible light. This shows the nucleus of the cells.

Green wavelength of light. No chemicals seemed to fluoresce with this wavelength.

Red wavelength of light. The cells fluoresce, indicating that there may be chlorophyll present in the cells.
So we finally have something to work off of - the potential presence of chlorophyll means that the cells would be photosynthetic. I had tried tackling the slime before with three separate 3-day lights outs periods with little success, it made a small dent in the slime but it always came back and my coral didn't really appreciate the low light period.
Knowing that the stuff is photosynthetic, a lights out period could harm it. How could I tackle it during the lights out period, while minimizing coral damage?
I first started by manually removing as much as I could, scrubbing every surface and scraping all of my glass. Normally when I do this, it comes back even bigger than before in a few days, but this I hoped teaming it up with a lights out period would stop it from growing back.
I then dosed #Vibrant - the liquid aquarium cleaner from @UWC. After manual removal and wrapping the tank, I dosed 1ml per 10g and waited three days.
I set my skimmer to skim VERY wet, it was pulling out a full cup every 12 hours. Something was happening, but what?
They were dying, that's what!
I think the combination of no light, manual removal getting most of the matter and Vibrant consuming the nutrients they were putting into the tank prevented them from coming back. It's been a week since I unwrapped the tank, and I've seen nary a trace. I'm still watching the tank like a hawk, but so far, it seems pretty good. I'm pretty happy with the results, and hopefully if someone out there is dealing with these, this post can help them out.
Thanks to everyone on R2R for the help in various threads, @UWC for the bacteria, and a huge thank you to Dr. Capman for the pictures and help identifying the slime.
















