- Joined
- Oct 15, 2019
- Messages
- 383
- Reaction score
- 1,600
Hi everyone,
I’m writing to bring to your attention a recent article entitled “Microbial Community Succession and Nutrient Cycling Responses following Perturbations of Experimental Saltwater Aquaria”. I don’t think we’ve discussed this study yet on R2R, but it’s directly relevant for many of the questions that are often brought up here. Fortunately this one is open-access, so you can read the whole thing here or download the PDF here.
This is the only peer-reviewed study of its kind that I know of, conducted by some of the leading experts in the use of these methods to study microbial communities. The first author, Holly Bik, has done a lot of really interesting work using DNA sequencing to study communities of various kinds. And the corresponding author, Jonathan Eisen, is a giant in this field who has played important roles in the Human Microbiome Project and other large microbiome projects.
The authors used DNA sequencing to study the microbial communities in aquarium water and surfaces (sound familiar?) They established and maintained a pair of large aquariums following what they deemed standard or best practices among aquarists. They sampled the microbial communities in these tanks repeatedly for about two months, to evaluate how the communities changed in response to actions by the aquarists.
Bik and coauthors found that
They found that the communities of microbes in saltwater aquariums remain relatively stable unless we do something specifically to alter them. This long stable period (from about Dec 15 to Jan 14) is readily visible in the following figure. And it shows that our actions, as aquarists, can have lasting effects on the microbiome of our tanks. See for example the dramatic changes in community composition after addition of live rock on Dec 12 and Jan 14 in this figure.
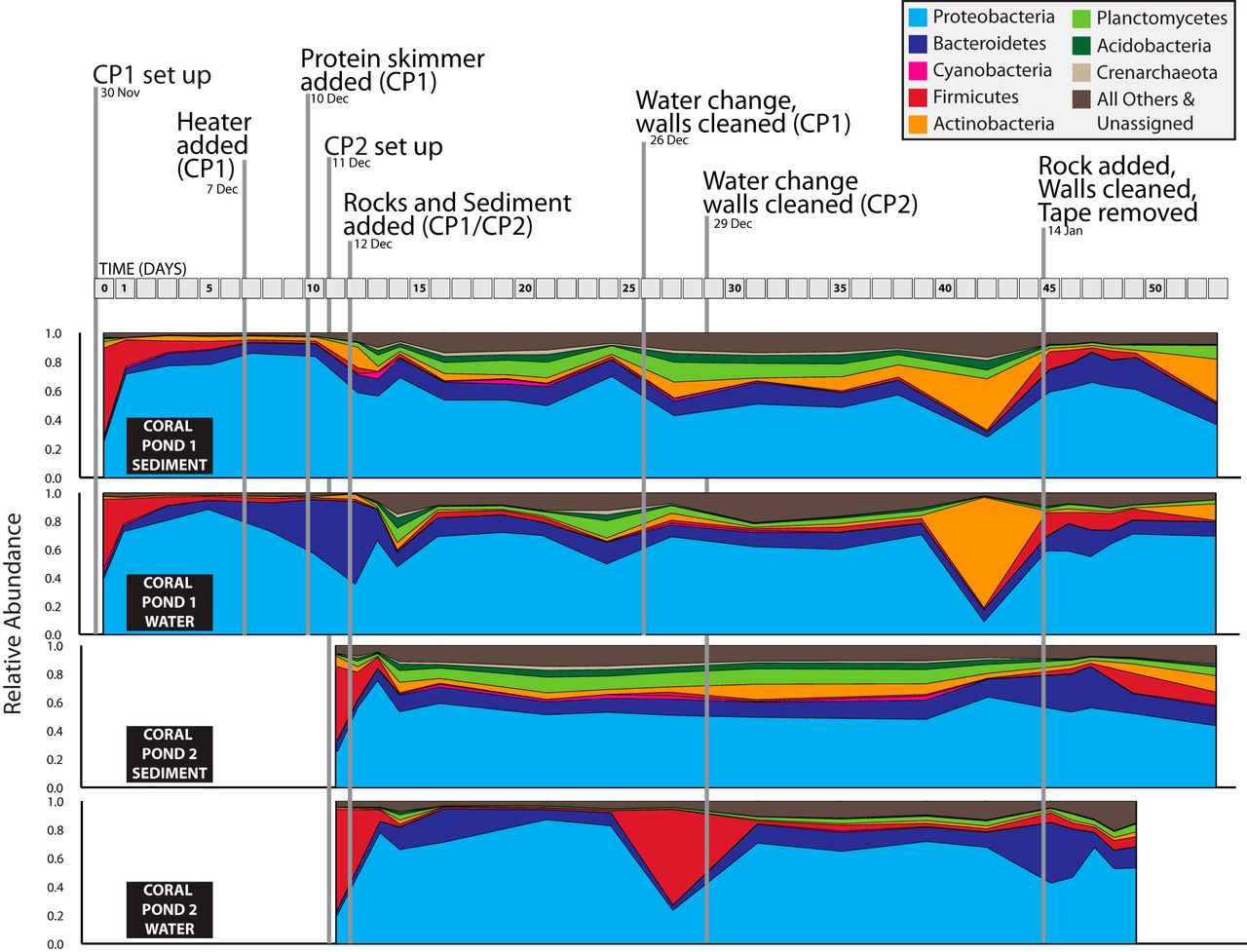
The most interesting part, for me, is the authors’ emphasis on “alternative stable states”. These communities generally remain stable, until disrupted by some action of the aquarist. Our actions can produce lasting changes in these communities, by shifting the community from one stable state to a different stable state. By repeatedly sampling a few tanks over the course of two months, the authors were able to observe the systems as they changed from one state to another.
This reminds me of the patterns I’ve described by surveying multiple hobbyist tanks. Aquarium microbiomes fell into a few clear clusters. These snapshots suggested that over time aquariums converged on one of a small number of patterns. Bik and her coauthors' study provides further support for this idea that there are alternative stable states in aquarium microbiomes.
One of these transitions (the first additional of live rock) clearly produced stable and beneficial changes in the microbial community. The second transition (additional live rock added at a later time) also affected the community, but the authors caution that (a) this transition was not accompanied by any measurable differences in function, and (b) further sampling would be needed to evaluate the stability of this second transition.
This later addition of live rock to an existing tank is something many people have asked me about. This study by Bik and coauthors shows evidence that this practice alters the microbial community. And I’ve recently described my own experience supplementing my home tanks with live sand and mud, showing that other (often much cheaper) materials can do similar things.
So the communities exist in a few different states, and we can shift the community between them depending on what we add, but the great question remains – what are the functional consequences of these different states? As the authors note at the end,
I will note here that the hobbyist community is conducting many thousands of experiments on this subject right now, with all the different products people are dosing in their tanks. The authors emphasize that we need to quantify the effects of these treatments. They only had two aquariums for this study, but with your (collectively) thousands of successful reef tanks, I think we’re in a good position to do exactly this. So let’s get to measuring!
I’m writing to bring to your attention a recent article entitled “Microbial Community Succession and Nutrient Cycling Responses following Perturbations of Experimental Saltwater Aquaria”. I don’t think we’ve discussed this study yet on R2R, but it’s directly relevant for many of the questions that are often brought up here. Fortunately this one is open-access, so you can read the whole thing here or download the PDF here.
This is the only peer-reviewed study of its kind that I know of, conducted by some of the leading experts in the use of these methods to study microbial communities. The first author, Holly Bik, has done a lot of really interesting work using DNA sequencing to study communities of various kinds. And the corresponding author, Jonathan Eisen, is a giant in this field who has played important roles in the Human Microbiome Project and other large microbiome projects.
The authors used DNA sequencing to study the microbial communities in aquarium water and surfaces (sound familiar?) They established and maintained a pair of large aquariums following what they deemed standard or best practices among aquarists. They sampled the microbial communities in these tanks repeatedly for about two months, to evaluate how the communities changed in response to actions by the aquarists.
Bik and coauthors found that
- Aquarium microbiomes remain generally stable during regular aquarium maintenance
- However, large disturbances can produce lasting changes in the community
- The addition of live rock rapidly produced diverse communities that resemble established aquariums
- This effect provided functional nitrifying communities at levels similar to established tanks
- Later addition of different live rock led to substantial changes in the microbiome
They found that the communities of microbes in saltwater aquariums remain relatively stable unless we do something specifically to alter them. This long stable period (from about Dec 15 to Jan 14) is readily visible in the following figure. And it shows that our actions, as aquarists, can have lasting effects on the microbiome of our tanks. See for example the dramatic changes in community composition after addition of live rock on Dec 12 and Jan 14 in this figure.

The most interesting part, for me, is the authors’ emphasis on “alternative stable states”. These communities generally remain stable, until disrupted by some action of the aquarist. Our actions can produce lasting changes in these communities, by shifting the community from one stable state to a different stable state. By repeatedly sampling a few tanks over the course of two months, the authors were able to observe the systems as they changed from one state to another.
This reminds me of the patterns I’ve described by surveying multiple hobbyist tanks. Aquarium microbiomes fell into a few clear clusters. These snapshots suggested that over time aquariums converged on one of a small number of patterns. Bik and her coauthors' study provides further support for this idea that there are alternative stable states in aquarium microbiomes.
One of these transitions (the first additional of live rock) clearly produced stable and beneficial changes in the microbial community. The second transition (additional live rock added at a later time) also affected the community, but the authors caution that (a) this transition was not accompanied by any measurable differences in function, and (b) further sampling would be needed to evaluate the stability of this second transition.
This later addition of live rock to an existing tank is something many people have asked me about. This study by Bik and coauthors shows evidence that this practice alters the microbial community. And I’ve recently described my own experience supplementing my home tanks with live sand and mud, showing that other (often much cheaper) materials can do similar things.
So the communities exist in a few different states, and we can shift the community between them depending on what we add, but the great question remains – what are the functional consequences of these different states? As the authors note at the end,
Bik et al. said:Future studies of aquaria should also … quantify the effects of specific perturbations. Aquaria represent an ideal mesocosm system that can be easily leveraged to test diverse biological hypotheses. (emphasis mine)
I will note here that the hobbyist community is conducting many thousands of experiments on this subject right now, with all the different products people are dosing in their tanks. The authors emphasize that we need to quantify the effects of these treatments. They only had two aquariums for this study, but with your (collectively) thousands of successful reef tanks, I think we’re in a good position to do exactly this. So let’s get to measuring!






















