Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
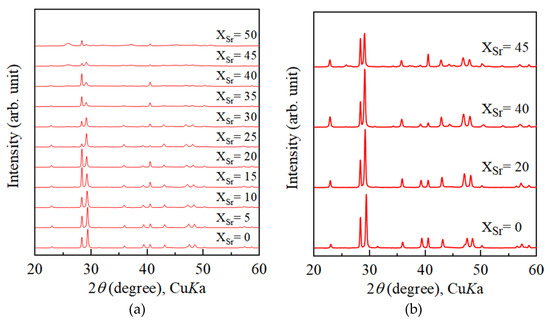
How about calcite?Magnesium and strontium are incorporated into deposited aragonite, even abiotic aragonite, in ratios that do not reflect the seawater concentration.
If I look at the figures in Hans-Werner:s paper - the ratio between Ca and Sr is nearly the same in both sea water and skeleton - around 50:1 What would happen if the ratio Ca/Sr in the tank water rise to 100:1 (1/2 natural Sr concentration). Would there still be the same ratio in the skeleton? - and if so - does it cost energy? If a biological process - it should IMO.
Sincerely Lasse