- Joined
- Oct 24, 2016
- Messages
- 1,547
- Reaction score
- 2,377
After manipulation and processing, these get very diluted down, which basically takes almost all the color with it as these serve such a small function in Vibrant as they do not have to have a strong presence. Totally opposite of a sludgy started bacterial product or any colored bacterial supplement in regards to this.Hi! I think I have polyglutamic acid, a similar anionic amino acid polymer. I can verify if this material can produce a color change. I highly doubt they will since both polyQ and polyD are both negatively charged, chemically similar and are unlikely to chelate the negatively charged dye. Heck, I'll even check some synthetic DNA today.
Nextly, I doubt there are cyanobacteria are in vibrant since the solution is clear and are not colored as cyanobacteria tend to be. In terms of bacillus, I guess they could be in there but the inactive spores of bacillus wouldn't be undergoing metabolism and probably wouldn't be producing these polyAAs, furthermore it wouldn't explain the algaecidal properties of vibrant since flocculants already exist for aquariums and they are not algaecide products. Although, to my understanding they could aid in the clearance of green water.
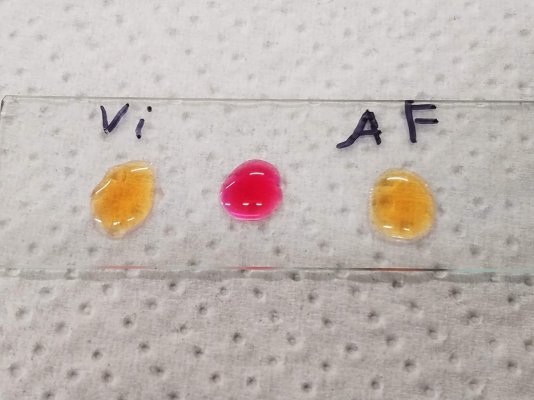
The pH of the BPB solution is controlled with excess phosphate buffer such that the pH contribution of any added analytes does not shift the color. Furthermore, the BPB colorshift to basic pH does not occur. @taricha has performed this test. Finally, the pH of both algaefix and vibrant are slightly acidic not basic.
In regards to your method, the method we both used was adapted from literature. But there are multiple ways of employing bromophenol blue to the detection of quaternary amines with or without chloroform extraction of the final complex. Basically as long as you add excess buffer and apply the solution to QAC, a color change occurs. No extraction is needed unless downstream analysis of the QAC is desired. OR if the analyte is in an unfriendly solution like milk. Considering that vibrant is clear as water. I don't think this is the case. Here are some literature (you can PM me if you'd like access):
Poly-γ-glutamate-based Materials for Multiple Infection Prophylaxis Possessing Versatile Coating Performance
Poly-γ-glutamate (PGA) possesses a nylon-like backbone and polyacrylate-like carboxyl groups, and shows an extraordinary solubility in water. In this study, the effective synthesis and structural analysis of some water-insoluble PGA ion-complexes (PGAICs) using cationic surfactants...www.mdpi.com

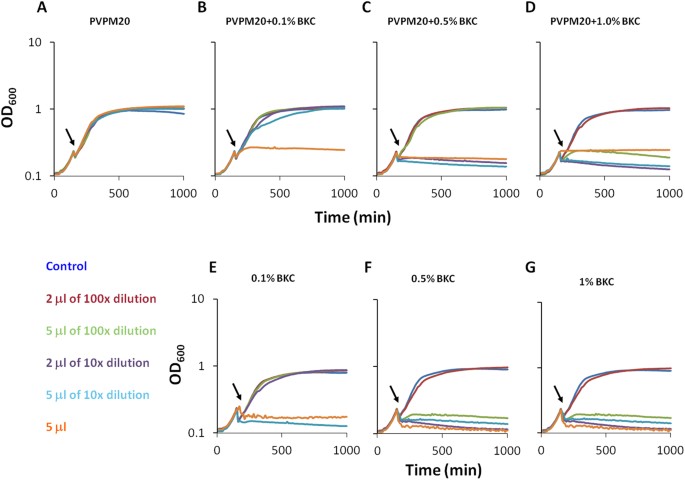
Colorimetric detection of residual quaternary ammonium compounds on dry surfaces and prediction of antimicrobial activity using bromophenol blue - PubMed
Controlling and monitoring the residual activity of quaternary ammonium compounds (QACs) are critical for maintaining safe yet effective levels of these agents in the environment. This study investigates the utility of bromophenol blue (BPB) as a safe, rapid and user-friendly indicator to detect...pubmed.ncbi.nlm.nih.gov
Towards an antimicrobial ‘microglove’ - Scientific Reports
A large proportion of hospital-related infections are acquired and spread due to the direct contacts between patients and healthcare workers. Accordingly, proper infection prevention measures and especially hand hygiene, are key to limit the spread of infections in nosocomial settings. However...www.nature.com
In the end. Vibrant likely contains quaternary ammonia or some other unlisted, highly charged compound capable of binding anionic dyes. Either way, its activity probably has nothing to do with anything listed on the label, in my opinion