There have been some discussion about using sodium hydroxide solution to remove CO2 in place of CO2 scrubber. So I actually tested it out.
The setup was simple. A media reactor, with the center pipe down to the bottom removed, with an air stone going in, feeded by an air pump (Whisper 10). I found in my previous test that putting input air into water column would cause too much back pressure to skimmer. So this time both input and output are above water, just letting the air stone to drive air through the solution to scrub out CO2.

The solution was prepared by adding 400g sodium hydroxide powder into 1300ml water.
When the air pump runs, it forms quite a bit of foam on top, probably because of the characteristic of sodium hydroxide solution. That caused some issue when the skimmer turned off (like during feeding), the bubble overflowed from the input hole, caused some mess.

Then the most important part, the effectiveness.
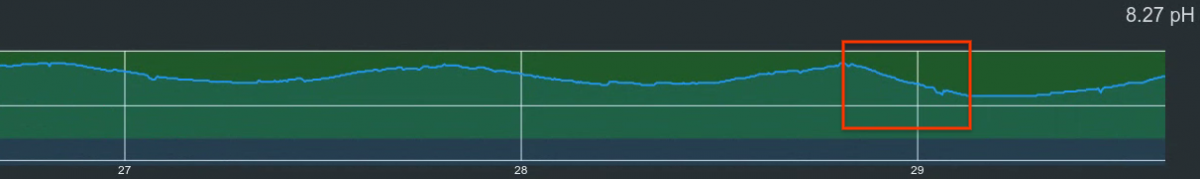
Here is the pH graph when I connected this reactor to the skimmer around dinner time. Light schedule is passed peak then. The pH soon started to drop in an increasing rate than it used to. By midnight, it dropped to 8.09. In the several nights before when the soda lime CO2 scrubber is used, the lowest it got is 8.21. At that point, I didn't believe it would slow down, so I unplugged the new reactor and plunged back in the typical one. pH flatlined right after. I think if I let it run overnight, the pH will drop below 8. I didn't want this trail become a stressful event for the tank. Also because of the overflow issue, I was not comfortable to leave it run overnight without oversight.
Although the test run is only lasted about 7 hours, I think it's enough to conclude that this design doesn't work. The effectiveness of it removing CO2 is far lower than soda lime. There are several possible reasons.
So overall, I think the idea of using sodium hydroxide as cheap alternative of soda lime to scrub CO2 in reef tank setting is not very practical.
The setup was simple. A media reactor, with the center pipe down to the bottom removed, with an air stone going in, feeded by an air pump (Whisper 10). I found in my previous test that putting input air into water column would cause too much back pressure to skimmer. So this time both input and output are above water, just letting the air stone to drive air through the solution to scrub out CO2.

The solution was prepared by adding 400g sodium hydroxide powder into 1300ml water.
When the air pump runs, it forms quite a bit of foam on top, probably because of the characteristic of sodium hydroxide solution. That caused some issue when the skimmer turned off (like during feeding), the bubble overflowed from the input hole, caused some mess.

Then the most important part, the effectiveness.

Here is the pH graph when I connected this reactor to the skimmer around dinner time. Light schedule is passed peak then. The pH soon started to drop in an increasing rate than it used to. By midnight, it dropped to 8.09. In the several nights before when the soda lime CO2 scrubber is used, the lowest it got is 8.21. At that point, I didn't believe it would slow down, so I unplugged the new reactor and plunged back in the typical one. pH flatlined right after. I think if I let it run overnight, the pH will drop below 8. I didn't want this trail become a stressful event for the tank. Also because of the overflow issue, I was not comfortable to leave it run overnight without oversight.
Although the test run is only lasted about 7 hours, I think it's enough to conclude that this design doesn't work. The effectiveness of it removing CO2 is far lower than soda lime. There are several possible reasons.
- the air pump output is not enough satisfy skimmer air input.
- the water volume is not deep enough, thus not enough contact time with the solution
- the bubble is too big, thus not enough contact surface area
So overall, I think the idea of using sodium hydroxide as cheap alternative of soda lime to scrub CO2 in reef tank setting is not very practical.




















