Okey, now I've been looking through old log files from 2013 to find some notes on a sulphur filter 
We put up a large DIY sulphur filter on a cold water system. Had the water in to the filter go through a DIY heat exchanger to get it up to 20-24 degrees, to speed up the bacterial growth.
Here are our notes. Water out from the filter:
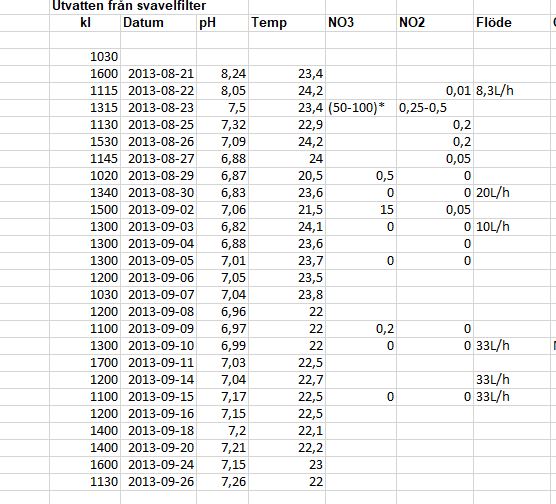
The pH is not totally correct. We didn't start with that high pH I think But you can see the drop in pH and approximately how much it decreased after the filter. I found notes from another sulphur filter we had, showing a pH at 6,8-7 in the water out.
But you can see the drop in pH and approximately how much it decreased after the filter. I found notes from another sulphur filter we had, showing a pH at 6,8-7 in the water out.
I didn't find that much on how much the KH increased after the coral gravel after the filter. That was with another filter, a couple of years later. But the reason we removed the coral gravel was that calcium got too high.
We put up a large DIY sulphur filter on a cold water system. Had the water in to the filter go through a DIY heat exchanger to get it up to 20-24 degrees, to speed up the bacterial growth.
Here are our notes. Water out from the filter:
The pH is not totally correct. We didn't start with that high pH I think
I didn't find that much on how much the KH increased after the coral gravel after the filter. That was with another filter, a couple of years later. But the reason we removed the coral gravel was that calcium got too high.


















