...deleted
Last edited:
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
For the moment, I'm going to sidestep the big question: whether having robust true Nitrification is actually important/beneficial in a system.
And say that if what you want is to establish a system with strong nitrification, it seems pretty clear to that for best results you've gotta be intentional about it.
Nitrifiers can be outcompeted for ammonia by coral, algae, and heterotrophs.
so...
No Light, no coral, no algae.
Start with ammonia dosing, not ghost feeding or a shrimp. Organics in the food will get the heterotrophs going, and the nitrifiers would only get ammonia as leftovers.
If you use a bottle starter, be sure it's true chemoautotroph nitrifiers. Some are actually "heterotrophic nitrifiers" that require Organic carbon. Adding ammonia-only, not a shrimp or fish food in the beginning will help distinguish.
Yeah I wouldn't go to that extent, just need to create the proper niche, so...For the moment, I'm going to sidestep the big question: whether having robust true Nitrification is actually important/beneficial in a system.
And say that if what you want is to establish a system with strong nitrification, it seems clear that for best results you've gotta be pretty intentional about it.
Nitrifiers can be outcompeted for ammonia by coral, algae, and heterotrophs.
so...
No Light, no coral, no algae.
Start with ammonia dosing, not ghost feeding or a shrimp. Organics in the food will get the heterotrophs going, and the nitrifiers would only get ammonia as leftovers.
If you use a bottle starter, be sure it's true chemoautotroph nitrifiers. Some are actually "heterotrophic nitrifiers" that require Organic carbon. Adding ammonia-only, not a shrimp or fish food in the beginning will help distinguish.
what is the benefit of adding that in addition to the live rock surface area
my thinking is the nh3 control is already permanently controlled even wo extra surface area
I don't see why not.So are you saying that if you started your tank as described above, you would have a system with initially strong nitrifiers?
So it wouldn't take many days at all (or many bottles) to scale up the nitrifiers as high as you could want.Out of 8 products tested i have found FRITZ TURBOSTART 900 ...normally in a 5 gal tank about 2 days with ammonia as high as 8-10ppm. Followed by Bio Spira and Dr Tim that took a day to two extra days to reduce/cycle tank fully
This is a really good question. I don't know. It would be cool if the answer was that the nitrifiers are pretty stable and aren't easily displaced from the sand. There are some reasons to think so, other reasons weigh against.What do you postulate will happen as the tank gets light, algae, CUC, pods, fish, corals, and generally matures? Would the nitrifiers lose out?
NH3 as a risk for livestock is of course permanently controlled in a mature system.my thinking is the nh3 control is already permanently controlled even wo extra surface area
If I hadn’t just recently watched 2 L of aerated tank water spiked with ammonia do nothing for several weeks, your results would have interested me not disturbed me
I am washing out flasks now to repeat your experiments.
I'll poke some more at your opinion that ammonia is a net positive later. I'm on the fence but slight lean against that.
My half-thought out argument is pretty simple. It revolves around location.Haha looking forward to that discussion. I'm going to come at it from the aspect of overall anabolism vs. catabolism and what it means in relation to keeping our likeable creatures happy while limiting pests.
@taricha here are the total ammonia concentrations for a sample of my aquarium water spiked with ammonium chloride over 48 hours. 30 mL samples were placed in glass stoppered 50 mL flasks which were placed on an orbital shaker in the dark. Triplicate measurements.
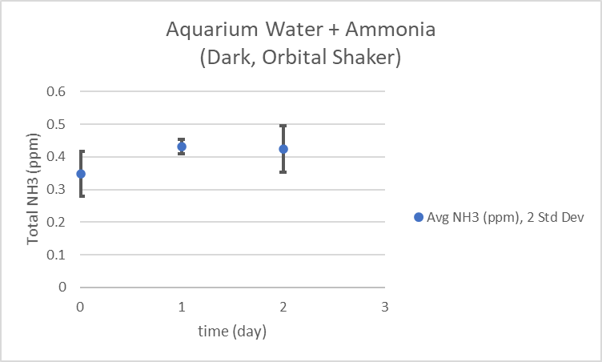
Since I put variable amounts of sand (0, 1, 2, or 5mL) in the same volume (100mL) of water moving the same way (orbital shaker), and I found that the amount of sand increased the ammonia consumed & NO3 produced, I have to conclude that it's mostly the amount of nitrifiers there controlling the rate in this test.Is it absent of nitrifiers or low in surface area and contact on only one presentation plane compared to live rock- can't move as much ammonia but still can move some into nitrate?
the water shear over just the top zone of sand, in eddy currents, seems like it would be less engaging than live rocks jutting mid water and all sides contact
Here's visualizing the rate of ammonia consumption (slopes from previous graph) vs the amount of sand there was.
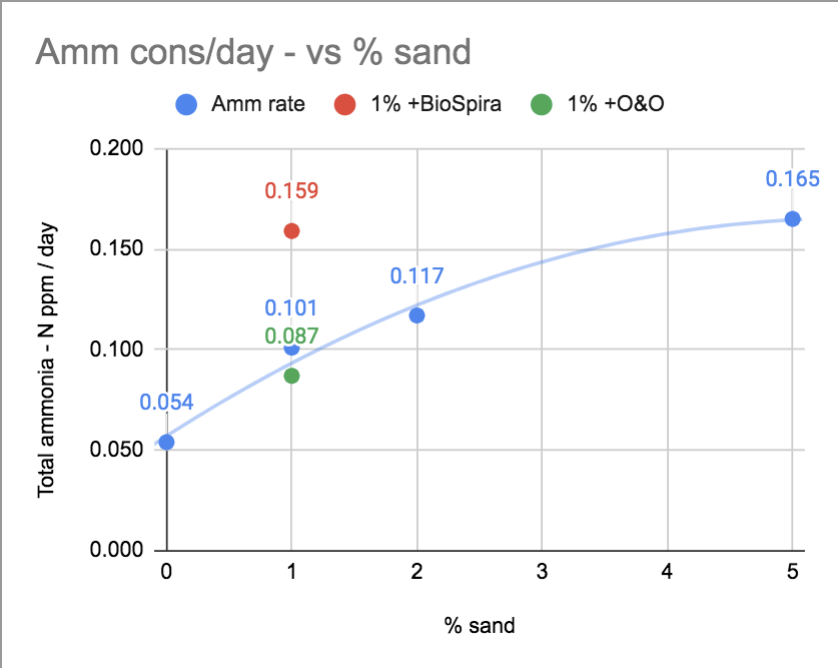
Well yes. Some. I'm definitely measuring some ammonia nitrification in the sand. It's just like 0.1 or 0.2ppm Total Ammonia-N per day compared to 20x-40x higher for my system overall.Surely something so in contact with the general tank has some nitrification ability, stuck to the sand grains?
Dan, Thanks so much for the legwork to attempt to replicate an unlikely result.
I have to suspect most people's water doesn't have a measurable appetite for ammonia. I won't really believe mine does either unless I see it again in a day or two, and even then - I'd feel a lot better if aquabiomics can find sufficient nitrifying microbes in the water to account for the result.
Big picture, what I found in the water was well smaller than what was in the sand - which itself was negligible compared to my overall system consumption of ammonia
Would you think that your aquarium water would show ammonia uptake? Or are the Pelagibacteraceae living in the sand doing their thing?FWIW: I don't want to make this about 'my tank', but here is an apparent example of the opposite of what you found sampling your sand and water. I thought this might be of interest as AquaBiomics produced an analysis of my 12g nano a while back:
"Your tank has nice high levels of nitrifying microbes. My view here is that a tank can process N through three different pathways (nitrification, heterotrophic assimilation, or photoautotrophic assimilation). In this context your tank appears to rely primarily on nitrification (rather than algal or heterotrophic uptake) to remove ammonia."
"Most tanks that have low scores have low Pelagibacteraceae, almost universally. Yours differs in the opposite direction -- your tank is dominated by this group. Pelagibacteracea are dominant in low-nutrient waters of the open ocean and is a major component of the typical reef tank. By having high levels, your tank is less like many reef tanks and more like natural reef waters."
From Wiki: Pelagibacteraceae ubique and related species are oligotrophs (scavengers) and feed on dissolved organic carbon and nitrogen.
Since I have very little visible algae, it seems that having a large percentage of nitrogen scavenging bacteria in the water column can contribute meaningfully to nitrogen processing. I just don't know the percentage breakdown for all the different pathways in the system.
No problem, though I admit the “Tom Sawyer painting the fence“ force is strong with you!Dan, Thanks so much for the legwork to attempt to replicate an unlikely result.
I have to suspect most people's water doesn't have a measurable appetite for ammonia. I won't really believe mine does either unless I see it again in a day or two, and even then - I'd feel a lot better if aquabiomics can find sufficient nitrifying microbes in the water to account for the result.
Big picture, what I found in the water was well smaller than what was in the sand - which itself was negligible compared to my overall system consumption of ammonia
Would you think that your aquarium water would show ammonia uptake? Or are the Pelagibacteraceae living in the sand doing their thing?
Interesting point about the potential interplay between Pelagibacteraceae and algae. I am studying biofilm formation in my system and looking for connections such as these.
So maybe a weak sandbed nitrifier population is a boon to nuisances on the substrate. And it's pretty clear that this weak nitrification in the sand would be very easy to fix.
FWIW: I don't want to make this about 'my tank', but here is an apparent example of the opposite of what you found sampling your sand and water. I thought this might be of interest as AquaBiomics produced an analysis of my 12g nano a while back:
"Your tank has nice high levels of nitrifying microbes. My view here is that a tank can process N through three different pathways (nitrification, heterotrophic assimilation, or photoautotrophic assimilation). In this context your tank appears to rely primarily on nitrification (rather than algal or heterotrophic uptake) to remove ammonia."
"Most tanks that have low scores have low Pelagibacteraceae, almost universally. Yours differs in the opposite direction -- your tank is dominated by this group. Pelagibacteracea are dominant in low-nutrient waters of the open ocean and is a major component of the typical reef tank. By having high levels, your tank is less like many reef tanks and more like natural reef waters."
From Wiki: Pelagibacteraceae ubique and related species are oligotrophs (scavengers) and feed on dissolved organic carbon and nitrogen.
Since I have very little visible algae, it seems that having a large percentage of nitrogen scavenging bacteria in the water column can contribute meaningfully to nitrogen processing. I just don't know the percentage breakdown for all the different pathways in the system.
Would you think that your aquarium water would show ammonia uptake? Or are the Pelagibacteraceae living in the sand doing their thing?
Interesting point about the potential interplay between Pelagibacteraceae and algae. I am studying biofilm formation in my system and looking for connections such as these.
Not so sure this would be easy to fix. Maintaining an aquarium with carbon dosing will necessarily favor heterotrophs. This includes utilizing algae for carbon dosing as they also increase overall DOC and POC levels.
If you want to maintain a larger nitrifying population (especially in areas with lower O2 percentage) you will need to decrease carbon input some how.
