Thank youVinegar contains acetic acid, which causes a portion of its pH drop instantly, and then less of a drop later when it is converted to CO2.
All of the pH lowering from CO2 comes later and more slowly with vodka.
Thus, folks can be rightfully concerned about the instantaneous pH drop when using vinegar in substantial amounts, but when slowly dosed, the pH effects ought to be similar for the same number of molecules dosed.
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Looking for thoughts on organic carbon dosing and nitrate
- Thread starter Randy Holmes-Farley
- Start date
- Tagged users None
@BaliReefBox see below - yes it did qualify for AA explains my fish behaviors!
I'm glad this thread was brought up because my 340g display defied logic. So, I believe there is something else at play. Scientists have complained that we need macro resources to study things at a large scale in the universe. It seems to me we need to study effects of carbon dosing on macro scales of aquariums. E.g. 500 gallon tanks, vs. 1000 gallon tanks, vs. 5000 gallon tanks.
Is there a ratio where carbon dosing has other unintended effects than reducing nitrate?
For me, my results were simple. I logged all my phosphate and nitrate Hanna tests in Apex while I was dosing straight vodka.
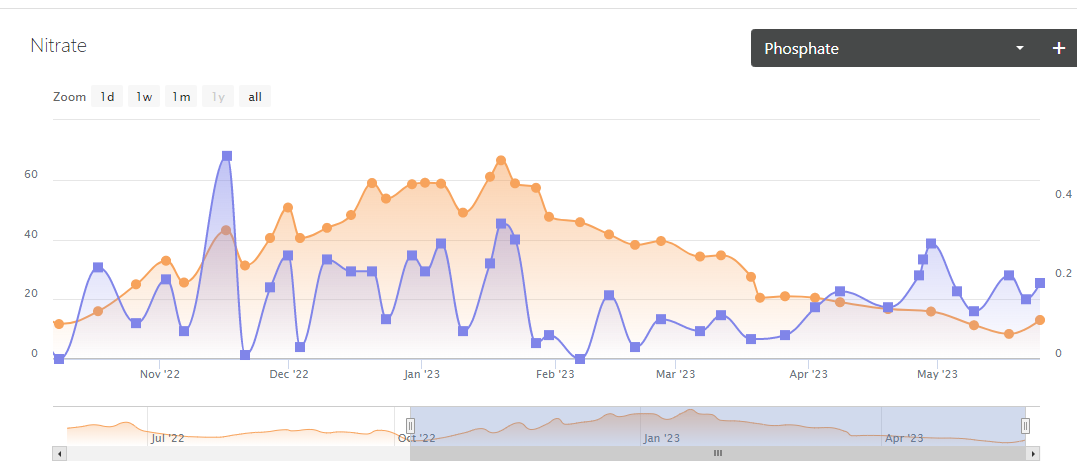
I'm pasting in a comparison chart of phosphates to nitrates since October of 2022 to current. I want you to guess when I stopped vodka dosing? I'll give one Hint I had started vodka dosing last spring about this time frame - May / June 2022.
***** Stop Reading Here to formulate your Guess as to when I stopped dosing******
5
4
3
2
1
I stopped dosing this past January. I was so fed up with nitrates and bacterial slime all over my tank. My gyres were constantly being gummed up by brown sludge. I had so much cloudy particulate in my 340 gallon display it was depressing to look at.
I stopped dosing vodka cold turkey. I did 2-3 50 % water changes between January and February. And you can see from January to Mid May my nitrates dropped steadily with no additional water changes since February. Also, no additional Vodka dosing. Once I got nitrates down from 65 ppm to under 35 ppm or so, something took off in the tank. Last week Nitrates hit there low point of 8 ppm. Afraid I'd bottom out I fed my old nitrate generating food. (Which I had stopped feeding last November / December). A couple feedings of that food and nitrates rose from 8 ppm to 13 ppm in a 1 week period.
My phosphates were all over the place. You can see when my nitrates hit 66 my phosphates peaked at .34 ppm. You can see in February my phosphates went from .3 down to .04 - .06 ppm down to 0.
I had phosphate present the majority of the time I was vodka dosing. At times very high phosphates.
Since stopping vodka dosing the results have been phenomenal over the last 4 months:
Coral Coloration has vastly improved
PH rose from a low of 7.6 - 7.7 to a low of 7.85 at night and 8.05 during the day
Cloudiness in the water has significantly reduced
Sludge on pumps has been eliminated
I didn't know why, but I had issues growing coraline algae on rocks and glass for the past 4 years. It's now taking off in a number of places.
I went from using over 110 inches of filter paper on my Reef Mat 1200 down to using 40-50 inches per day! Saving me $35 + 2 bottles of vodka a month = $55-$60 a month in savings)
The difference between when I was dosing and now, is night and day. People wouldn't recognize the tank.
Some things to note: I ran all of the below while carbon dosing
I do run a skimmer
I do run a Reefmat
I do run an algae turf scrubber (which was producing massive amounts of algae weekly)
I do run a fluidized pellet reactor with pellets that break down to do carbon dosing
I do run a Nu-Clear Cannister filter - Cartridge removed full of Matrix rock and Aqua Char
At my peak I was dosing 65 ml of vodka per day - I used a dosing pump on a timer and dosed 4-5 times a day.
I don't know if it took 6-8 months for bacteria to build up on the matrix rock while carbon dosing, if it took a year for the pellet reactor to kick in. But I credit my success now to those two things running in tandem and removing the vodka dosing all together. I don't know if there was competition. That while vodka dosing the pellets and the matrix rock were competing and dying causing a build up of nitrates instead of consumption? The output of the matrix rock and the pellet reator, 1 goes to my skimmer intake, and the other goes to the beginning of my sump. They are both ran off my return pump.
I feel like my case, is epidemy of failed vodka dosing. My Nitrates sky rocketed while dosing. . .
I'm not a vodka advocate by no means but there are so many variables in your case that makes the conclusion that it was the vodka causing problems you had not really plausible.
You had vodka, pellets, algae scrubber, skimmer, reef mat, canister filter running and you had problems. At some point you decided to stop one of them (in this case vodka dosing) and to do 2-3 very big 50% water changes. That improved the things drastically and you decided it was vodka causing all your problems. But how do you know? If happened by chance to stop for ex pellets, or algae scrubber, but not vodka, and then do this big water changes, how do you know there wont be similar improvement?
I would say the big water changes (2-3 50%) in short time were the game changer in your case and removing one (by chance vodka) of so many (6! - almost all possible) nutrient reduction methods helped you to solve your problems.
I didn't go into the history of it all but, you're right, when one thing didn't seem to have an impact I added another. I bought the $300 (with pellets) fluidized reactor thinking this would solve all my nitrate problems. The vendor assured me it would in 6-8 weeks if I dosed bacteria like microbacter clean or mb 7. I dosed clean according to the directions and the vendor's directions for the pellet reactor. I dosed daily (manually) and after 8 weeks my nitrates were still rising with no visible changes to anything. At the time I was using the 25 micron cannister filter and decided to swap it out with matrix rock. After all, maybe the pellets just weren't enough. So, I did that. I dosed MB Clean for another 2-3 weeks and waiting a full 8 weeks for both, thinking that at some point things are just going to take off and nitrates will crash (that was my biggest fear at the time).I'm not a vodka advocate by no means but there are so many variables in your case that makes the conclusion that it was the vodka causing problems you had not really plausible.
You had vodka, pellets, algae scrubber, skimmer, reef mat, canister filter running and you had problems. At some point you decided to stop one of them (in this case vodka dosing) and to do 2-3 very big 50% water changes. That improved the things drastically and you decided it was vodka causing all your problems. But how do you know? If happened by chance to stop for ex pellets, or algae scrubber, but not vodka, and then do this big water changes, how do you know there wont be similar improvement?
I would say the big water changes (2-3 50%) in short time were the game changer in your case and removing one (by chance vodka) of so many (6! - almost all possible) nutrient reduction methods helped you to solve your problems.
I was wrong... After 4 months of the pellet reactor running and the 2 months of the matrix rock reactor running, nitrates continued to climb. I decided to start dosing vodka. I started at 10 ml per day for 2 weeks, then worked my way up to 40 ml per day. At the time I saw Nitrates stop climbing and stabilize. They stayed the same week after week of testing. I thought o.k. something is working. After two months of Vodka dosing, 4 months of the matrix rock in place and 6 months of the pelletized reactor, I thought something maybe started working. So, I raised the vodka dosing to 50 ml per week for 2 weeks. Then 60ml for a couple months.
PH kept decreasing, corals stopped growing, things just didn't look healthy. Nothing died perse, but things didn't look good either. Color was being lost, etc. I reduced vodka dosing back down to 40 ml per day and said screw it, I don't know what to do, I'll just let it run for another few months. Sometimes no intervention is better. Over that time I continued to test weekly, but, things didn't change. Phosphates continued to fluctuate up and down week to week. One week they'd be zero, the next week they'd be as high as .2. Nitrates continued to fluctuate in the 40s for months.
Then, I decided to take a metaphorical wrecking hammer to it all. It was a year of pent up reef frustration. PH wouldn't rise no matter what I did, I tried a CO2 scrubber, I tried moving my skimmer airhose intake outside, I tried a lot. PH would increase by maybe .1. So, after a year's worth of fighting it, I decided, it's time for water changes. I thought I'll just do one 40-50% water change and leave it at that. Well, I wasn't o.k. with that. I did at least one more, I might have done a third, about 2 weeks a part each. I tested before and after to ensure it had the mathematical impact I expected. And it worked. The last water change , nitrates tested at 23 ppm. I also stopped vodka dosing then, and then shouted some swear words at the bacteria in my tank and walked away.
I didn't really want to test again, I expected within a few days nitrates would skyrocket and I'd be back to square one. But, I made myself test. There wasn't huge move and it was well within the margin of error but that first test a couple weeks after the third water change came in at 17ppm. I was happy if it just stayed there. I'd been elated.
Then I tested again a week later, and they were down to 13 outside the margin of error. The next test they were down to 8.
I agree with you there a lot of changes, however, this has been an 18 month battle with nitrates, ph, and getting things to be managed.
What do I think actually happened? I sound crazy for saying this, but, what I actually think happened is high nitrates limit denitrifying bacteria.
I don't know the science so, it's just a guess, but the directions on the Matrix rock (SeaChem) and BRS Bio Pellets, both said the exact same thing. Nitrates need to be under 20 ppm for these to work. (Probably not in those words). I added both when Nitrates were between 40 and 65 and waited half a year for them to do something. It could be just concidence that the water changes knocking nitrates down to the 20s did something, or it allowed the media to take off. I don't understand why that would be.
I wouldn't even know how to test that? Is there some inverse relationship between carbon dosing and too much food for bacterial strains, or can carbon fuel other types of bacterial strains that are less efficient in higher nitrate environments? Can Nitrate limit what the bacteria can accomplish?
Who knows what is in the pellets and how it will interact with ethanol. If there are two of you who experience problems when combining this two carbon sources I suppose there is a problem. But I would not blame vodka method or pellet method both of them work for many people.I didn't go into the history of it all but, you're right, when one thing didn't seem to have an impact I added another. I bought the $300 (with pellets) fluidized reactor thinking this would solve all my nitrate problems. The vendor assured me it would in 6-8 weeks if I dosed bacteria like microbacter clean or mb 7. I dosed clean according to the directions and the vendor's directions for the pellet reactor. I dosed daily (manually) and after 8 weeks my nitrates were still rising with no visible changes to anything. At the time I was using the 25 micron cannister filter and decided to swap it out with matrix rock. After all, maybe the pellets just weren't enough. So, I did that. I dosed MB Clean for another 2-3 weeks and waiting a full 8 weeks for both, thinking that at some point things are just going to take off and nitrates will crash (that was my biggest fear at the time).
I was wrong... After 4 months of the pellet reactor running and the 2 months of the matrix rock reactor running, nitrates continued to climb. I decided to start dosing vodka. I started at 10 ml per day for 2 weeks, then worked my way up to 40 ml per day. At the time I saw Nitrates stop climbing and stabilize. They stayed the same week after week of testing. I thought o.k. something is working. After two months of Vodka dosing, 4 months of the matrix rock in place and 6 months of the pelletized reactor, I thought something maybe started working. So, I raised the vodka dosing to 50 ml per week for 2 weeks. Then 60ml for a couple months.
PH kept decreasing, corals stopped growing, things just didn't look healthy. Nothing died perse, but things didn't look good either. Color was being lost, etc. I reduced vodka dosing back down to 40 ml per day and said screw it, I don't know what to do, I'll just let it run for another few months. Sometimes no intervention is better. Over that time I continued to test weekly, but, things didn't change. Phosphates continued to fluctuate up and down week to week. One week they'd be zero, the next week they'd be as high as .2. Nitrates continued to fluctuate in the 40s for months.
Then, I decided to take a metaphorical wrecking hammer to it all. It was a year of pent up reef frustration. PH wouldn't rise no matter what I did, I tried a CO2 scrubber, I tried moving my skimmer airhose intake outside, I tried a lot. PH would increase by maybe .1. So, after a year's worth of fighting it, I decided, it's time for water changes. I thought I'll just do one 40-50% water change and leave it at that. Well, I wasn't o.k. with that. I did at least one more, I might have done a third, about 2 weeks a part each. I tested before and after to ensure it had the mathematical impact I expected. And it worked. The last water change , nitrates tested at 23 ppm. I also stopped vodka dosing then, and then shouted some swear words at the bacteria in my tank and walked away.
I didn't really want to test again, I expected within a few days nitrates would skyrocket and I'd be back to square one. But, I made myself test. There wasn't huge move and it was well within the margin of error but that first test a couple weeks after the third water change came in at 17ppm. I was happy if it just stayed there. I'd been elated.
Then I tested again a week later, and they were down to 13 outside the margin of error. The next test they were down to 8.
I agree with you there a lot of changes, however, this has been an 18 month battle with nitrates, ph, and getting things to be managed.
What do I think actually happened? I sound crazy for saying this, but, what I actually think happened is high nitrates limit denitrifying bacteria.
I don't know the science so, it's just a guess, but the directions on the Matrix rock (SeaChem) and BRS Bio Pellets, both said the exact same thing. Nitrates need to be under 20 ppm for these to work. (Probably not in those words). I added both when Nitrates were between 40 and 65 and waited half a year for them to do something. It could be just concidence that the water changes knocking nitrates down to the 20s did something, or it allowed the media to take off. I don't understand why that would be.
I wouldn't even know how to test that? Is there some inverse relationship between carbon dosing and too much food for bacterial strains, or can carbon fuel other types of bacterial strains that are less efficient in higher nitrate environments? Can Nitrate limit what the bacteria can accomplish?
After so many interventions and mixture of methods nobody can say what was wrong with your aquarium.
I would avoid general conclusions about the effectiveness of any of methods you were using. I'm pretty sure you were mad on pellets and 300$ reactor few months ago and was blaming this method also not working at all. But today (after you lower nitrates to the levels required by the producer) you are very happy with the results it provides
Enjoy your nice tank, I am pretty sure you learned your lessons.
IMO - this happens all the time when different organics (organic detritus) is broken down by bacteria. Its not only a stockpile for N - it will also release P. . .
. . . Note - P can only come into the system as an import that we bring in - food, introduced organisms that die and so on. It can not be created internally from nothing. . . .
Depending on what's deposited in carbonate substrates endoliths will release P as they dissolve the substrates. Sponges sequester polyphosphate crystals. It's unclear which sponge species in our aquaria do this and to what extent but sponges samples I've had tested when a friend's system had issues maintianing PO4 had P crystals imbeded in their tissue. While sponges can be shedding large amounts of N rich detritus the PolyP may be retained. De Goiej pointed out it's not just bacteria that is breaking down detritus but filter feeders and macro organisms are also feeding on the sponge detritus.
The release of ammonia and/or nitrate from organisms consuming detritus is presumably happening all the time, and the nitrate concentration is the result of many processes, including the one you mention adding to it.
If the organic carbon dosing causes bacteria to take up nitrate, then nitrate will decline unless you are proposing that the worms etc. eating detritus become more active with organic carbon dosing/nitrate decline, and do so to a degree to offset the nitrate taken up by bacteria. That last part is what does not seem likely to me.
Remember, sponges may be taking up labile DOC 1000X faster than the bacterioplabnkton. Certainly bacteria will be taking up nitrate but how much DOC are they actually getting initially? With the N rich detritus from sponges starting to show up in the food web in just 2 days we're looking at what seems to me a signifiacant adjsutment in the food web in a very short time.
Another way the food webs in aquaria is likely to be altered is individual corals reaction to the growth of heterotrophic bacteria in their individual microbiomes or surface mucus layers. With the research showing labile DOC can create create anoxic or hypoxic conditions for corals would certainly disrupt their uptake of nitrogen compounds.
An update. From yesterday nitrate started rising - because it is Salifert test was not sure yesterday but today is clear - it is between 2.5 and 5ppm.I've started a simple experiment to dose organic carbon some two weeks ago exactly because of this thread, so thank you Randy.
This tank is new - about 4 months old, 26 gal about 20 gal water. 5 small fish, cuc, cleaner shrimp. LPS corals, trumpets, torch, favia, duncan, goni, and a easy SPS - stylophora "Milka" Feeding 1 cube frosen artemia daily. Live rocks, sand. Never dosed bacteria. Started N-cycle with few drops of mixture of ammonium, nitrate, urea and aminoacids (leftovers from a old experiment). Lights at full duration of 10 hours from the day one - Radion 15. Filtration Tunze 9004 skimmer, no sump, no mechanical filtration. There is some detritus build up already maybe because of lack of additional filtration.
Pests: Only one rock covered with turf algae, (why they are only on this rock is another story, maybe good to start new build thread)
Normal calcium, magnesium, alkalinity levels.
Dosing calcium formate 5 ml/day, and about 0.05 -0.1 ppm phosphate daily. I am dosing phosphate because of that rock with turf algae - I have a Lawnmower Blenny and I like him very much, great character, but he is eating only algae. Actually I think he hates algae as much as regular reefer do, because after he eats a lot of them making himself look as a fat tadpole he starts just scrubbing them out making my life harder to clean powerhead and skimmer from algae on a daily basis.
When started experiment NO3 was above10 ppm (Salifert), PO4 undetectable by Salifert (despite dosing - new sand, you know).
Started with 1 ml /day DIY version of NOPOX - ethanol, methanol, acetic acid, isopropanol, this time added to the recipe about 30 gram per liter glucose. Mixing nopox with 1 ml calcium carbonate prior dosing. Dosing after light is off. I have continued daily dosing of PO4 about 0.1 ppm when the light is on. The water became slightly cloudy for 2-3 days (not because of calcium carbonate) and on glass appeared whitish green slime, I decided to leave it like it is. The skimmer air intake clogged with bacterial buildup. No problems for fish or corals all of them were happy and breathing normal.
I have clearly noticed nitrate reduction already on the day 3 - it was between 5-10 ppm. At the day 5-7 it was about 2.5 - 5 ppm. At this point turf algae became bright green, started growing very fast, and started spreading on the rocks nearby, on the bottom appeared brown large spots of dino (Amphidinuim - checked under mic).
At this stage decided to only stop dosing PO4 and reduce the light duration from 10 to 8 hours. Continued dosing 1 ml/ day Nopox. Feeding the same 1 cube of brine shrimp. Nitrate reduction stopped at 2.5 ppm.
Today is day 14 of carbon dosing and Nitrate stays at about 2.5 ppm (salifert) for more than 7 days. What changed in the tank? Well, turf algae (and Blenny) are facing really hard times turf algae very noticeably declined for just one week, dinos are barely visible only for 1-2 hours during the light is on. And it looks the build up detritus on the rocks and bed is visually less now.
All the coral, fish and other inhabitants are happy (maybe except Blenny but he still have enough dying algae to eat).
I'll continue dosing 1 ml/day for another week and will report back.
it was flatline at 2.5 for about 12 days. I am still dosing 1 ml NoPOX and trace metals daily (about 20 gal water).
There is no big change with the rock with turf algae since last time. Detritus build-up is gone, dinos also. there is no clouding after dosing. Water is clear. No noticeable bacterial growth on substrate rock glass, only skimmer is clogging more frequently. There is increased volume of the skimmate. This evening skimmate turned green which in my experience is bad signal, I have noticed some of the candy corals are starting losing tissue from the base.
At this stage I have several options:
1. Stop the experiment
2. Continue without changes
3. Resume phosphate dosing without other changes
4. Halve carbon dosing and resume dosing phosphate
5. Stop carbon dosing and resume phosphate dosing
What is your opinion?
Edit: (The aim of this simple experiment was to see how fast the system will respond to carbon dosing in terms of nitrate reduction)
Last edited:
I would start dosing PO4 again + lowering DOC with 50 %
Sincerely Lasse
Sincerely Lasse
Here is a living example of what I´m trying to say - its from my own aquaria. I dose ethanol 8% in the plenum of my reversed flow DSB (deep gravel filter). Because of my own mistake - I got a lot of DOC coming out in the DT and nearly kill some of my LPS (bacteria growth) I had to stop the DOC dosing for 6 days. - What happen´s - Right - NO3 rise from around 3 to nearly 24 mg/L, Start dosing again - NO3 after 7 days - 9 mg/L. Today I dose 1.8 ml/2 hour
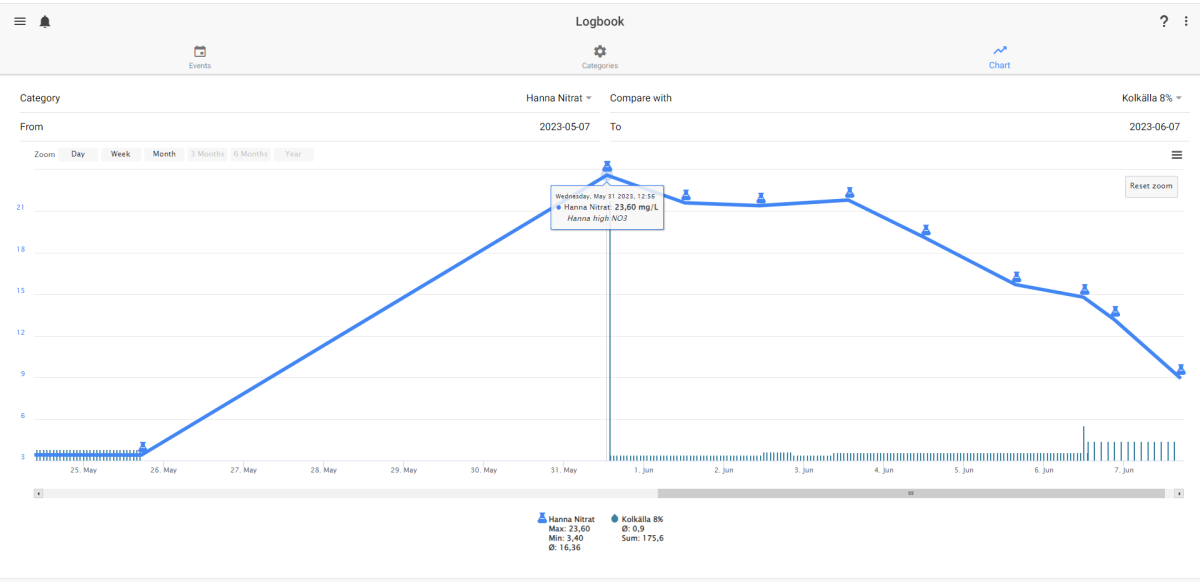
This is an example on DOC´s importance for denitrification. At the same time PO4 gone from 0.23 mg/L to 0.07 mg/L. However it is not depending of DOC dose. It was 0.23 before I stop the DOC and still 0.23 when I start DOC 6 days later. But at the same time I start the DOC dosing - I changed my GFO to new. And if the PO4 decrease was due to biomass growth - it should only be responsible for a NO3 removal of 2 mg NO3/L (max)
Sincerely Lasse
This is an example on DOC´s importance for denitrification. At the same time PO4 gone from 0.23 mg/L to 0.07 mg/L. However it is not depending of DOC dose. It was 0.23 before I stop the DOC and still 0.23 when I start DOC 6 days later. But at the same time I start the DOC dosing - I changed my GFO to new. And if the PO4 decrease was due to biomass growth - it should only be responsible for a NO3 removal of 2 mg NO3/L (max)
Sincerely Lasse
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,349
- Reaction score
- 63,689
Here is a living example of what I´m trying to say - its from my own aquaria. I dose ethanol 8% in the plenum of my reversed flow DSB (deep gravel filter). Because of my own mistake - I got a lot of DOC coming out in the DT and nearly kill some of my LPS (bacteria growth) I had to stop the DOC dosing for 6 days. - What happen´s - Right - NO3 rise from around 3 to nearly 24 mg/L, Start dosing again - NO3 after 7 days - 9 mg/L. Today I dose 1.8 ml/2 hour

This is an example on DOC´s importance for denitrification. At the same time PO4 gone from 0.23 mg/L to 0.07 mg/L. However it is not depending of DOC dose. It was 0.23 before I stop the DOC and still 0.23 when I start DOC 6 days later. But at the same time I start the DOC dosing - I changed my GFO to new. And if the PO4 decrease was due to biomass growth - it should only be responsible for a NO3 removal of 2 mg NO3/L (max)
Sincerely Lasse
Adding organic matter directly into a plenum will, IMO, hugely increase the percentage of organic added that is used for denitrification, and presumably that is why you dose that way.
I'd be wary of extrapolating to aquaria adding organics to the water column however. I've always been skeptical of achieving rapid denitrification when one needs all of:
1. organics to diffuse more rapidly into the sand or rock than O2,
2. nitrate to diffuse more rapidly than O2
3. organics to diffuse about as fast as nitrate (or faster).
Any organic we use should diffuse more slowly than O2.
I was dosing vinegar for the first time... plenty of nitrates (due to accidental over-dosing of about 100ppm), plenty of phosphates (0.2ppm).. Following the accident I have been dosing vinegar for 3 weeks and it does not really seem to have much of an impact on my tank nitrate levels (other than what I would see as a natural reduction over time due to refugium and live rock).
Before the accident, I had 0 nitrates and dosed ~3ppm per day to keep it slightly above zero (so that's at least ~20ppm per week consumption)..
Therefore for me (it seems) - carbon dosing simply does nothing.. I can see the impact of vinegar dosing on tank PH (slightly lower perhaps 0.2PH lower) but that's it...
Also, FYI - I am running SPS only tank (only have 1 accidental LPS - hammer) and I was worried that it will negatively impact my stony corals... How wrong I was! Nothing was at all bothered that nitrates shot 100ppm within a 5 min time span and that it stayed elevated for weeks!
Before the accident, I had 0 nitrates and dosed ~3ppm per day to keep it slightly above zero (so that's at least ~20ppm per week consumption)..
Therefore for me (it seems) - carbon dosing simply does nothing.. I can see the impact of vinegar dosing on tank PH (slightly lower perhaps 0.2PH lower) but that's it...
Also, FYI - I am running SPS only tank (only have 1 accidental LPS - hammer) and I was worried that it will negatively impact my stony corals... How wrong I was! Nothing was at all bothered that nitrates shot 100ppm within a 5 min time span and that it stayed elevated for weeks!
Last edited:
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,349
- Reaction score
- 63,689
I was dosing vinegar for the first time... plenty of nitrates (due to accidental over-dosing of about 100ppm), plenty of phosphates (0.2ppm).. Following the accident I have been dosing vinegar for 3 weeks and it does not really seem to have much of an impact on my tank nitrate levels (other than what I would see as a natural reduction over time due to refugium and live rock).
Before the accident, I had 0 nitrates and dosed ~3ppm per day to keep it slightly above zero (so that's at least ~20ppm per week consumption)..
Therefore for me (it seems) - carbon dosing simply does nothing.. I can see the impact of vinegar dosing on tank PH (slightly lower perhaps 0.2PH lower) but that's it...
How much are you dosing to what size tank?
I have a relatively small 4 foot tank (Reefer 425XL), currently dosing around 30ml of distilled malt vinegar per day (started with 15 ml and increased to 30ml over a week or so).. I think this is ballpark what you and others recommended on a per gallon basis previously..How much are you dosing to what size tank?
I´m not sure that I follow you. IMO - More DOC - more consumption of O2. In the sediment DOC increase the bacterial growth and consume O2 in the first step. When O2 is gone att x µm in the biofilm/sediment NO3 start to be consumed instead.. Because DOC is the limited growth factor - it will be rapidly consumed in the interface between water and sediment - no need for diffusing further down. However - your are right - you can´t expect a rapid denitrification in these cases but you will have it sooner or later. This is exactly my point - when you after a certain time note a decrease in NO3 (higher than before) - the most likely reason is denitrification not growthI've always been skeptical of achieving rapid denitrification when one needs all of:
1. organics to diffuse more rapidly into the sand or rock than O2,
2. nitrate to diffuse more rapidly than O2
3. organics to diffuse about as fast as nitrate (or faster).
Any organic we use should diffuse more slowly than O2.
Edit: By the way - in nitrifying biofilms it had been shown that O2 levels are low already after some µm and heterotrophic bacterial growth are ten fold higher - if there is DOC enough - the O2 will disappear fast.
Sincerely Lasse
Last edited:
Post cycle my nitrates were over 80 ppm and couldn’t lower them with NoPox until I added phosphates. Redfield ratio seems to not exist in an aquarium yet I believe both nitrogen and phosphorus needs to exist to reduce either based on my experience. Purely anecdotal and could just be a coincidence but it’s worked for me since. Have no issues bottoming nitrates out and getting phosphates low requiring something such as lanthanum chloride to bottom the other in my tests. Perhaps bottoming nitrates out impeded the other coming down naturally or not. Just believe both must exist. To what extent each needs the other I don’t know.I was dosing vinegar for the first time... plenty of nitrates (due to accidental over-dosing of about 100ppm), plenty of phosphates (0.2ppm).. Following the accident I have been dosing vinegar for 3 weeks and it does not really seem to have much of an impact on my tank nitrate levels (other than what I would see as a natural reduction over time due to refugium and live rock).
Before the accident, I had 0 nitrates and dosed ~3ppm per day to keep it slightly above zero (so that's at least ~20ppm per week consumption)..
Therefore for me (it seems) - carbon dosing simply does nothing.. I can see the impact of vinegar dosing on tank PH (slightly lower perhaps 0.2PH lower) but that's it...
Also, FYI - I am running SPS only tank (only have 1 accidental LPS - hammer) and I was worried that it will negatively impact my stony corals... How wrong I was! Nothing was at all bothered that nitrates shot 100ppm within a 5 min time span and that it stayed elevated for weeks!
Last edited:
- Joined
- Sep 21, 2018
- Messages
- 6,675
- Reaction score
- 7,170
How much vinegar are you dosing per gallon of water in the system?I was dosing vinegar for the first time... plenty of nitrates (due to accidental over-dosing of about 100ppm), plenty of phosphates (0.2ppm).. Following the accident I have been dosing vinegar for 3 weeks and it does not really seem to have much of an impact on my tank nitrate levels (other than what I would see as a natural reduction over time due to refugium and live rock).
Before the accident, I had 0 nitrates and dosed ~3ppm per day to keep it slightly above zero (so that's at least ~20ppm per week consumption)..
Therefore for me (it seems) - carbon dosing simply does nothing.. I can see the impact of vinegar dosing on tank PH (slightly lower perhaps 0.2PH lower) but that's it...
Also, FYI - I am running SPS only tank (only have 1 accidental LPS - hammer) and I was worried that it will negatively impact my stony corals... How wrong I was! Nothing was at all bothered that nitrates shot 100ppm within a 5 min time span and that it stayed elevated for weeks!
- Joined
- Sep 21, 2018
- Messages
- 6,675
- Reaction score
- 7,170
Is this about 100 gallons? If so, you are only 1/3 the way to a sufficient dose, though nitrate reduction could occur below 1 mL per gallon.I have a relatively small 4 foot tank (Reefer 425XL), currently dosing around 30ml of distilled malt vinegar per day (started with 15 ml and increased to 30ml over a week or so).. I think this is ballpark what you and others recommended on a per gallon basis previously..
Reefer XL425 is 88 gal so maybe water is 80 gal? But still half of the dose. And also there are so many kinds of vinegar with different strength. Not sure about strength of "distilled" malt vinegar.Is this about 100 gallons? If so, you are only 1/3 the way to a sufficient dose, though nitrate reduction could occur below 1 mL per gallon.
Lasse can you provide some references on this. Very interesting.Edit: By the way - in nitrifying biofilms it had been shown that O2 levels are low already after some µm and heterotrophic bacterial growth are ten fold higher - if there is DOC enough - the O2 will disappear fast.
Similar threads
- Replies
- 10
- Views
- 388
- Replies
- 184
- Views
- 11,032
- Replies
- 14
- Views
- 319
- Replies
- 41
- Views
- 1,895
-
- AMS: Article
- Replies
- 287
- Views
- 12,844
New Posts
-
-
-
Frag Build It was this big... Now its this big...
- Latest: Reefing_addiction