Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
When and why did dosing nitrates start?
- Thread starter 2Sunny
- Start date
- Tagged users None
Okay so exogenous ammonia is used by most organisms on the planet and exogenous ammonia is released by most organisms on the planet. And exogenous ammonia is also synthesized from N2 and hydrogen and released by nitrogen fixing prokaryotes. And DNRA prokaryotes take nitrate and produce and release ammonia. So not sure where you're going with this. I suggest you take a look at my thread in my signature and also read my article when it comes out.
Last edited:
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
I must have express myself in a bad way - IMO - they form mats when there is low or no inorganic N and/or low PO4 in the water. They need to get the nutrients from another wayHowever where is the evidence that cyano form mats without amino acids? It is much more likely they form mats when ammonia is not available.
Sincerely Lasse
- Joined
- Sep 21, 2018
- Messages
- 9,785
- Reaction score
- 9,639
The point is that heterotrophic organisms need a source of organic carbon to generate energy and if there are other atoms attached, such as N and P, it does not need, theses elements become waste. Because organisms do not perform atom accounting as they bring molecules into the cytoplasm, they end up needing to the throw away unneeded molecules, such as ammonia and phosphate. Given that available food is rarely atom balanced and heterotrophs need to generate energy by converting organic carbon to carbon dioxide, ammonia and phosphate must be eliminated. This is easily demonstrated by aging aquarium water a few days with a little fish food.In all living cells, the universal energy currency is adenosine triphosphate, or ATP. The 3 phosphates are where electrons come from to provide energy for cellular processes. The enzymes driving all of this are proteins containing N, which lower energy requirements thus enabling reactions to proceed, which might not otherwise.
To be accurate, all 3 elements C, N P & many others also, play crucial roles in driving cellular energy transfer. I wouldn’t exactly call N & P leftovers.
- Joined
- Sep 21, 2018
- Messages
- 9,785
- Reaction score
- 9,639
If sufficient nitrogen, phosphorous and a pinch of other things are not available, biomass, i.e. mats, cannot form. Both viewpoints need to be rethought, for example, organic matter. Lasse, I am still working on a rebuttal to your theory.I must have express myself in a bad way - IMO - they form mats when there is low or no inorganic N and/or low PO4 in the water. They need to get the nutrients from another way
Sincerely Lasse
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
Exactly - they have to seek it in another place when the left over in the water column gone away. In sediments or in organic matter. But it cost energy and it is easier to get P and N in the water column but if it is empty there - they form mats and get their nutrients from sediments (even inorganic) and from organic matter . The mats - the secret is mostly hydrocarbons from the photosynthesis - giving carbon to the bacteria working below the mats and to the cyanobacteria IMO. The mat forming bacteria is always present - the mats not. If you can repeat your test with PO4 and minerals in the water but only get mat forming when you ad amino acids - you have got something - but use F/2 solution this time (with and without inorganic N)If sufficient nitrogen, phosphorous and a pinch of other things are not available, biomass, i.e. mats, cannot form.
A good sign that it takes time :DLasse, I am still working on a rebuttal to your theory.
Sincerely Lasse
- Joined
- May 22, 2016
- Messages
- 7,223
- Reaction score
- 10,586
This is a great discussion, here's another illustration of some of what's being discussed - here's what can happen when aquarium heterotrophic bacteria get fed some amino acid.
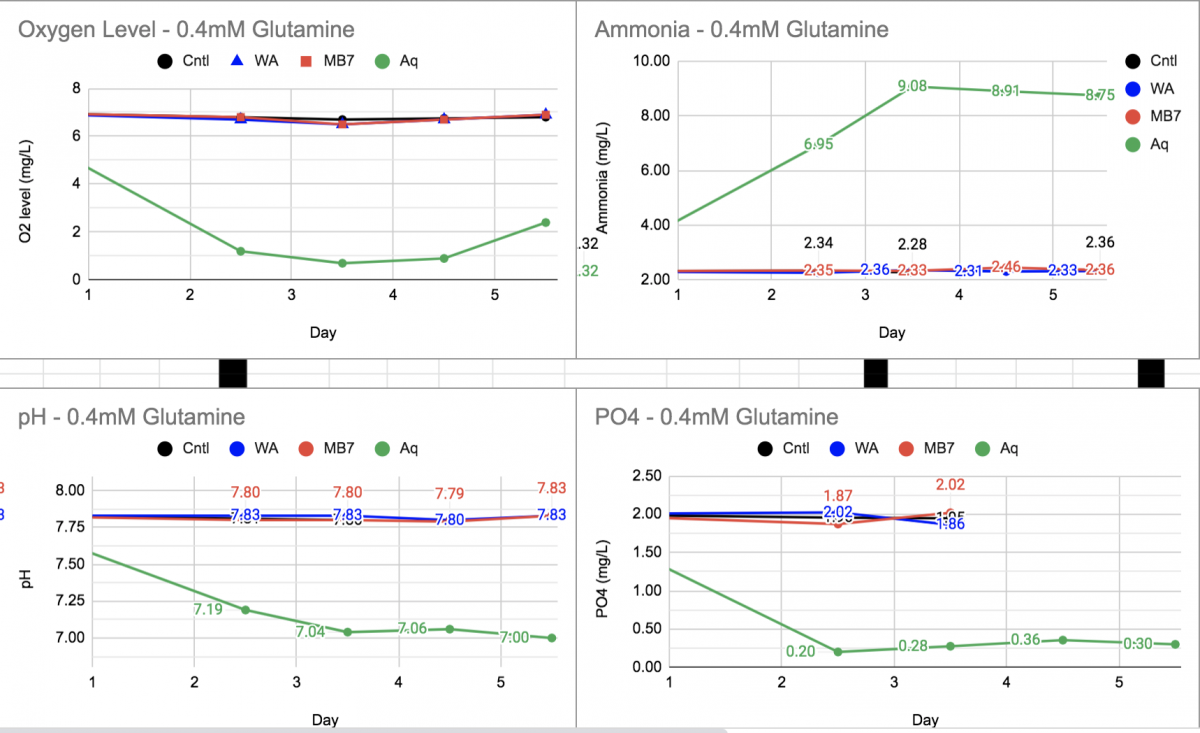
"Aq" in green is random aquarium bacteria from the water, "Cntl" in black got nothing and should have stayed sterile, and the Red and blue are other bacterial sources that didn't want to activate (but that's another topic).
Top left, O2 is consumed - in this case enough to essentially deplete the O2 in the container. The O2 consumption is from the carbon in the amino acid being oxidized to CO2 for energy.
Top right, Ammonia is produced gradually for 3.5 days. This is connected to the O2 consumption. When bacteria burn the carbon in the amino acid for energy, the N in the amino acid gets released. Based on the amount of ammonia increase, ~7ppm and the total amount of N added in glutamine - enough to produce ~14ppm ammonia, in this case about half the N went into biomass, and the other half was released as ammonia.
bottom left: pH drop in the closed container likely due to CO2 production (but maybe other acids also?).
bottom right: PO4 decreased as the bacteria took it in to build biomass.
Just another illustration of what's being said in this good discussion regarding the mechanics of feeding amino acids (organic nitrogen-carbon mix) in our tanks.
"Aq" in green is random aquarium bacteria from the water, "Cntl" in black got nothing and should have stayed sterile, and the Red and blue are other bacterial sources that didn't want to activate (but that's another topic).
Top left, O2 is consumed - in this case enough to essentially deplete the O2 in the container. The O2 consumption is from the carbon in the amino acid being oxidized to CO2 for energy.
Top right, Ammonia is produced gradually for 3.5 days. This is connected to the O2 consumption. When bacteria burn the carbon in the amino acid for energy, the N in the amino acid gets released. Based on the amount of ammonia increase, ~7ppm and the total amount of N added in glutamine - enough to produce ~14ppm ammonia, in this case about half the N went into biomass, and the other half was released as ammonia.
bottom left: pH drop in the closed container likely due to CO2 production (but maybe other acids also?).
bottom right: PO4 decreased as the bacteria took it in to build biomass.
Just another illustration of what's being said in this good discussion regarding the mechanics of feeding amino acids (organic nitrogen-carbon mix) in our tanks.
I like to thank you all for taking the time and giving these detailed responses. I am learning a lot.
This is a great discussion, here's another illustration of some of what's being discussed - here's what can happen when aquarium heterotrophic bacteria get fed some amino acid.

"Aq" in green is random aquarium bacteria from the water, "Cntl" in black got nothing and should have stayed sterile, and the Red and blue are other bacterial sources that didn't want to activate (but that's another topic).
Top left, O2 is consumed - in this case enough to essentially deplete the O2 in the container. The O2 consumption is from the carbon in the amino acid being oxidized to CO2 for energy.
Top right, Ammonia is produced gradually for 3.5 days. This is connected to the O2 consumption. When bacteria burn the carbon in the amino acid for energy, the N in the amino acid gets released. Based on the amount of ammonia increase, ~7ppm and the total amount of N added in glutamine - enough to produce ~14ppm ammonia, in this case about half the N went into biomass, and the other half was released as ammonia.
bottom left: pH drop in the closed container likely due to CO2 production (but maybe other acids also?).
bottom right: PO4 decreased as the bacteria took it in to build biomass.
Just another illustration of what's being said in this good discussion regarding the mechanics of feeding amino acids (organic nitrogen-carbon mix) in our tanks.
The drop in phosphate of ~75% is very telling & appears to match O2 consumption due to respiration.
The drop in pH and rise in ammonia match as well. When ammonia is dissolved in water it begins to ionize to NH4+, and more so at higher pH. As NH4+ is oxidized, H+ ions are released, causing a drop in pH. Studies have shown that pH shift can cause a decrease in biodiversity in soils. Whether true in an aquatic environment or not, I’m not sure?
Also, if I remember correctly, the initial oxidation of ammonium to nitrite is rate limited, so might explain the temporal differences observed.
I will take some time to digest the stoichiometric implications. Thank you for sharing the data.
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
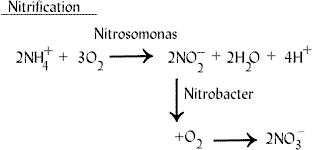
My boldWhen ammonia is dissolved in water it begins to ionize to NH4+, and more so at higher pH
In fact it is the total opposite
Here is a very good (and famous) diagram showing that
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
I´m sorry - but it going better - it is halfway to be correct :D:D:D Each NH4 will donate 2 H+ith the oxidation of NH4+ comes 4 H+ ions.
Sincerely Lasse
Last edited:
Brew12
Electrical Gru
View BadgesExcellence Award
Reef Tank 365
Article Contributor
Moderator Emeritus
North Alabama Reef Club
Article Administrator
My Tank Thread
I know... I know.. I should be learning about biomass, carbon, amino acids and other aquatic concepts. Instead, I can't help but be amazed you are having this conversation in something other than your native language. Well done!I´m sorry - but it going better - it is halfway to be correct :D:D:D On each NH4 will it be 2 H+
Sincerely Lasse
Ok.. you can go back to these high level discussions now.
So, I am seeing numerous accomplished reefers offering advice to beginners to "get their nitrates up" or to even dose nitrates.
Everywhere you look people are cramming their tanks full of slick hip hi-tech nutrient export gear and filtration. I'm convinced mainly because it looks slick hip and hi-tech in Instagram and Facebook photos. Why? Because everyone else does and newer reefers, and slick, hip, hi-tech social media types feel the need to compete in the battle of slick hip hi-tech reef tanks.
Just a hunch.
- Joined
- May 22, 2016
- Messages
- 7,223
- Reaction score
- 10,586
To be clear, in my samples like these (a bottle with a little aquarium water added to inoculate), any signs of nitrification - ammonia decrease and Nitrite production - have taken 20-25 days to show up. So none happening in the data posted above over 6 days.the initial oxidation of ammonium to nitrite
Dang, I think Randy HF is deep into the bag of popcorn watching this one! Some heavy stuff going on here, but I am gleaning good bits that address the original question.
This is a great discussion, here's another illustration of some of what's being discussed - here's what can happen when aquarium heterotrophic bacteria get fed some amino acid.

"Aq" in green is random aquarium bacteria from the water, "Cntl" in black got nothing and should have stayed sterile, and the Red and blue are other bacterial sources that didn't want to activate (but that's another topic).
Top left, O2 is consumed - in this case enough to essentially deplete the O2 in the container. The O2 consumption is from the carbon in the amino acid being oxidized to CO2 for energy.
Top right, Ammonia is produced gradually for 3.5 days. This is connected to the O2 consumption. When bacteria burn the carbon in the amino acid for energy, the N in the amino acid gets released. Based on the amount of ammonia increase, ~7ppm and the total amount of N added in glutamine - enough to produce ~14ppm ammonia, in this case about half the N went into biomass, and the other half was released as ammonia.
bottom left: pH drop in the closed container likely due to CO2 production (but maybe other acids also?).
bottom right: PO4 decreased as the bacteria took it in to build biomass.
Just another illustration of what's being said in this good discussion regarding the mechanics of feeding amino acids (organic nitrogen-carbon mix) in our tanks.
Good stuff @taricha just one minor quible. The O2 is being reduced to water. The CO2 is from the oxidation of the carbon of the glutamine.
Likely pH drop from CO2 production and fermentation products such as lactate, acetate, etc
Or, it could be - that's the advice the reefing community has been giving people for 30 years.Everywhere you look people are cramming their tanks full of slick hip hi-tech nutrient export gear and filtration. I'm convinced mainly because it looks slick hip and hi-tech in Instagram and Facebook photos. Why? Because everyone else does and newer reefers, and slick, hip, hi-tech social media types feel the need to compete in the battle of slick hip hi-tech reef tanks.
Just a hunch.
You can look at a thread on RC from the early 2000s and the advice we give people is exactly the same as it was then - despite equipment being drastically better than it was.
Buy a skimmer rated for way bigger than your tank. Frequent large water changes. Big refugiums. Undetectable nitrates and phosphates, Feed less, Etc.
Every problem newbies have - the solution they're given is to drive nutrients lower.
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
@ichthyoid Here is a part about alkalinity and nitrification - source here
Sincerely Lasse
- Alkalinity and pH: Alkalinity is lost in an activated sludge process during nitrification. Nitrifiers use alkalinity as a carbon source, i.e., they use an inorganic form of carbon. Hydrogen ions (H+) are produced when ammonium ions are oxidized to nitrite: NH4+ + 1.5O2 → 2H+ + NO2- + 2H2O. Nitrous acid (HNO2) is also produced during the oxidation of ammonium ions. This destroys alkalinity: H+ + NO2- → HNO2. 7.14 mg of alkalinity as CaCO3 are destroyed for every mg of ammonium ions oxidized. If the pH drops below 6.7, there is a significant decrease in nitrification. Therefore, it is important to maintain an adequate alkalinity in the aeration tank to provide pH stability and also to provide inorganic carbon for nitrifiers. After complete nitrification, a residual alkalinity of 50 mg/l in the aeration tank is desirable. If this alkalinity is not present, then alkalinity should be added to the aeration tank. The optimal pH range for nitrification is 7.2 to 8.0. A substantial reduction in nitrification activity occurs at pH levels below 6.7.
Sincerely Lasse
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
And the funny results is that it is more problem with Cyanobacteria and dinoflagellates today compared with 30 years ago - and we keep up the same mantra.Every problem newbies have - the solution they're given is to drive nutrients lower.
Sincerely Lasse
Similar threads
- Replies
- 3
- Views
- 882
- Replies
- 27
- Views
- 993
- Replies
- 9
- Views
- 290
- Replies
- 4
- Views
- 2,301
TOP 10 Trending Threads
-
- Question
- Replies
- 22
- Views
- 413
- Replies
- 67
- Views
- 1,615
-
- Sticky
- Replies
- 95
- Views
- 962
- Replies
- 23
- Views
- 419
- Replies
- 57
- Views
- 724
- Replies
- 61
- Views
- 1,445
- Replies
- 27
- Views
- 506
- Replies
- 32
- Views
- 686
- Replies
- 40
- Views
- 1,378
New Posts
-
-
if you could say one thing to online vendors, what would it be?
- Latest: christinna77

















