- Joined
- Sep 21, 2018
- Messages
- 6,690
- Reaction score
- 7,180
Recently, I spent time in the lab looking at why so much water is needed to rinse the fines from new dry aragonite sand (Caribsea). I blame @brandon429 for such an odd pursuit. Fines are the very small bits of sand that can cloud aquarium water for days when they are not rinsed from the sand. Our current method of washing a bag of sand is to place it in a bucket, add water, swirl the mixture, pour off the cloudy water, add more water, repeat and repeat and repeat and repeat. What I found explains why we have to repeat the rinse cycle so many times: we don’t mix the water-sand mixture vigorously or long enough.
Fines seem to tenaciously adhere to larger sand grains and a minimum amount of force is needed to knock them loose. Swirling a sand-water mixture for a short while delivers only enough energy to knock off a small portion of the fines. This is one reason why our sand rinse method requires protracted swirling. The reason for the large amount of water use is caused by the assumption that once the water becomes cloudy, it must be replaced with clean water to continue fines removal. Under laboratory conditions, most of the fines in a batch of sand can be suspended in one volume of wash water. In the plot below, data from lab mixing experiments demonstrate this.
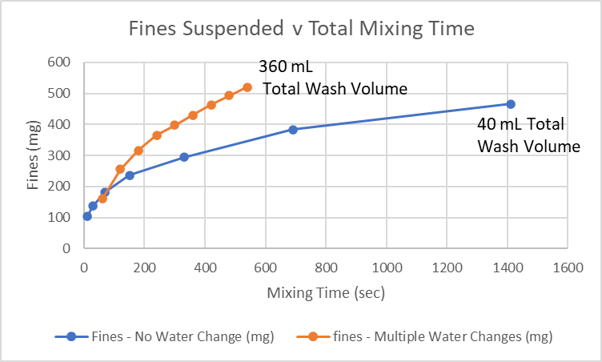
In the first experiment, 20 g of sand was repeatedly rinsed with clean water. Each rinse used 40 mL of water and was mixed for one minute with a 30 watt vortex mixer. This represents the standard sand wash procedure. The orange line shows the sum of fines suspended over time. A little over 500 mg was removed with 360 mL of water. The experiment was then repeated but the rinse water was not changed but mixed until roughly 500 mg of fines were suspended. Only 40 mL of water were used. Clean water does seem to play a beneficial role in fines removal because less than 80% of the fines are removed for the same total mixing time in the second experiment. After obtaining these results, I wondered whether an outboard motor would be needed to stir a bag of sand and water enough to duplicate these 20 g experimental results.
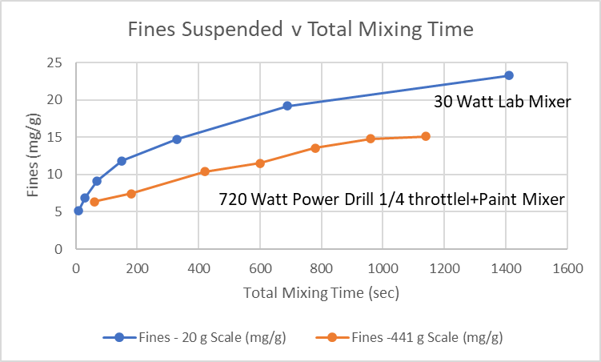
My only scale up experiment was 441 g of sand and 900 mL water, only a small portion of a bag of sand but large enough to provide a hint of the power needed to mix a large batch. For mixing I used a power drill fitted with a paint mixer (see photo).

As with the 20 g experiment, the wash water was not changed. The experiment was conducted in an open 2.5 gallon bucket, which limited how fast the mixture could be stirred without ejecting it from the bucket. The plot below compares the relative effectiveness of fines suspension for the vortex mixer with 20 g of sand and the power drill with 441 g. The large scale experiment suspended less than 75% of the fines for the same amount of time. I suspect going to a covered bucket and the drill at full speed might have brought the large scale results closer. These result do confirm that a full bag of sand would require longer mixing time with a power drill if an outboard motor is unavailable.
These results are not sufficient to provide specific recommendations on the amount of water and mixing time needed for rinsing a bag of sand, though I would say you probably cannot over mix a sand-water mixture and you will be able to cut way back on rinse water amount.
Fines seem to tenaciously adhere to larger sand grains and a minimum amount of force is needed to knock them loose. Swirling a sand-water mixture for a short while delivers only enough energy to knock off a small portion of the fines. This is one reason why our sand rinse method requires protracted swirling. The reason for the large amount of water use is caused by the assumption that once the water becomes cloudy, it must be replaced with clean water to continue fines removal. Under laboratory conditions, most of the fines in a batch of sand can be suspended in one volume of wash water. In the plot below, data from lab mixing experiments demonstrate this.
In the first experiment, 20 g of sand was repeatedly rinsed with clean water. Each rinse used 40 mL of water and was mixed for one minute with a 30 watt vortex mixer. This represents the standard sand wash procedure. The orange line shows the sum of fines suspended over time. A little over 500 mg was removed with 360 mL of water. The experiment was then repeated but the rinse water was not changed but mixed until roughly 500 mg of fines were suspended. Only 40 mL of water were used. Clean water does seem to play a beneficial role in fines removal because less than 80% of the fines are removed for the same total mixing time in the second experiment. After obtaining these results, I wondered whether an outboard motor would be needed to stir a bag of sand and water enough to duplicate these 20 g experimental results.
My only scale up experiment was 441 g of sand and 900 mL water, only a small portion of a bag of sand but large enough to provide a hint of the power needed to mix a large batch. For mixing I used a power drill fitted with a paint mixer (see photo).
As with the 20 g experiment, the wash water was not changed. The experiment was conducted in an open 2.5 gallon bucket, which limited how fast the mixture could be stirred without ejecting it from the bucket. The plot below compares the relative effectiveness of fines suspension for the vortex mixer with 20 g of sand and the power drill with 441 g. The large scale experiment suspended less than 75% of the fines for the same amount of time. I suspect going to a covered bucket and the drill at full speed might have brought the large scale results closer. These result do confirm that a full bag of sand would require longer mixing time with a power drill if an outboard motor is unavailable.
These results are not sufficient to provide specific recommendations on the amount of water and mixing time needed for rinsing a bag of sand, though I would say you probably cannot over mix a sand-water mixture and you will be able to cut way back on rinse water amount.
















