So I've been in discussions with people about this, and I feel like I need to better understand the issue.
So below summarises my understanding of the issue, based on what I've read and heard from the likes of @Lou Ekus . I could be wrong!
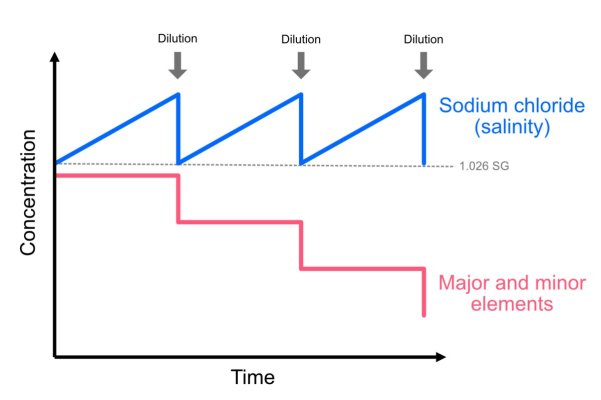
In the figure below, I am hypothetically dosing a 2-part solution that adds additional sodium chloride (blue) to the tank, which is an unavoidable byproduct of using sodium-based carbonates and calcium chloride additives.
Over time, the sodium chloride builds up and increases salinity, but my other major and minor trace elements are (initially) at the right levels (red line). When I dilute my tank to bring the salinity down, I reduce the sodium chloride levels, but inadvertently, I'm also diluting the other elements in the tank. Over a long enough time period, this results in depleted levels of major and trace elements, even though the sodium chloride level is staying stable.
This is the current issue I am experiencing, due to the fact I dose a fair amount of 2-part on a relatively small amount of water (due to high coral biomass). About once a month, even with water changes, I need to make a correction to my salinity, and it is unavoidable that this will lower other elements.
With a part C, which is sodium chloride free saltwater, additional major and minor trace elements are added, so they build up in higher concentrations, in the same ratio as the sodium chloride. When the water is then diluted to bring salinity down, everything is diluted back down to the optimal level.
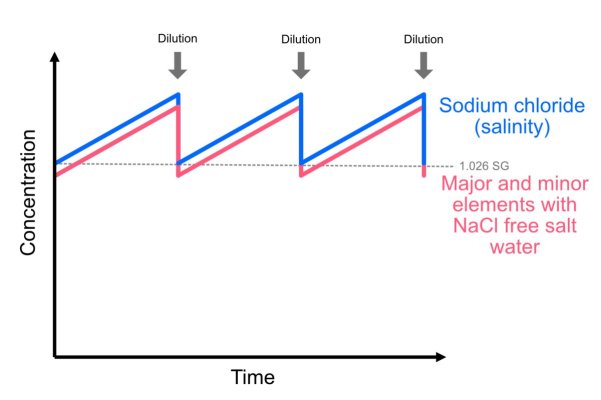
(1) Firstly, is this understanding correct? So for example, additional calcium is being added over time, and without dilution it would actually be overdosed, in the same was sodium chloride is effectively being over dosed?
(2) Secondly, why doesn't a 2-part that contains additional traces solve this issue? My understanding is that the traces are not being overdosed, but rather "matched" to alkalinity consumption.
I'd really like to nail my comprehension of this issue down, so thanks in advance for any input!
So below summarises my understanding of the issue, based on what I've read and heard from the likes of @Lou Ekus . I could be wrong!
In the figure below, I am hypothetically dosing a 2-part solution that adds additional sodium chloride (blue) to the tank, which is an unavoidable byproduct of using sodium-based carbonates and calcium chloride additives.
Over time, the sodium chloride builds up and increases salinity, but my other major and minor trace elements are (initially) at the right levels (red line). When I dilute my tank to bring the salinity down, I reduce the sodium chloride levels, but inadvertently, I'm also diluting the other elements in the tank. Over a long enough time period, this results in depleted levels of major and trace elements, even though the sodium chloride level is staying stable.
This is the current issue I am experiencing, due to the fact I dose a fair amount of 2-part on a relatively small amount of water (due to high coral biomass). About once a month, even with water changes, I need to make a correction to my salinity, and it is unavoidable that this will lower other elements.

With a part C, which is sodium chloride free saltwater, additional major and minor trace elements are added, so they build up in higher concentrations, in the same ratio as the sodium chloride. When the water is then diluted to bring salinity down, everything is diluted back down to the optimal level.

(1) Firstly, is this understanding correct? So for example, additional calcium is being added over time, and without dilution it would actually be overdosed, in the same was sodium chloride is effectively being over dosed?
(2) Secondly, why doesn't a 2-part that contains additional traces solve this issue? My understanding is that the traces are not being overdosed, but rather "matched" to alkalinity consumption.
I'd really like to nail my comprehension of this issue down, so thanks in advance for any input!














