Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Low Ph - How concerned should I be?
- Thread starter R33fDaddy
- Start date
- Tagged users None
lets slow this runaway wagon down just a bit. when people talk about chasing numbers, this is a prime example of it. the situation that you are running in to is that you have two test methods that are giving you different results. one a manual test, and the otehr is an equipment test. your ph has reflected great on the manual test, and the probe test is not, and you have decided to disregard the manual test you did, for the sake of assuming that the probe test is the accurate of the two, and now youre buying scrubbers, buffers, etc to correct a number from a probe, that may not be showing right.
everythign from maybe buying plants and whatnot to fix pH that may not need to be fixed, if your manual testing shows a good pH. if thats the case, i have a bridge to sell you.
everythign from maybe buying plants and whatnot to fix pH that may not need to be fixed, if your manual testing shows a good pH. if thats the case, i have a bridge to sell you.
as far as the apex system, one thing to note is that there are a couple things that can affect the results that you get for readings. one of them is that it doesnt read right, straight out of the box. it might take a couple days, as far as what i saw from other users, as well as for myself. in addition to that, the probe wires are said to be affected by power current ie: a power cord too close to the probe wire. ive also heard alot of people having issues with probe readings due to bubbles on the sensor of the probe. heck, it could be a sensor issue just in itself.
if you have multiple manual tests that show a number consistently, but the pH probe is the only thing that isnt reflecting the same, then i would work out the probe issue before you start throwing fixes at your tank. you very well could be doing stuff to fix an issue that isnt an issue at all.
if you have multiple manual tests that show a number consistently, but the pH probe is the only thing that isnt reflecting the same, then i would work out the probe issue before you start throwing fixes at your tank. you very well could be doing stuff to fix an issue that isnt an issue at all.
I get what you're saying, but it's actually more to it. I have a pink/yellow and Red Gonipora that had been closed up for for weeks. What I didn't post was that once I got the CO2 Scrubber the very next day the pink one was fully extended. The Red one has started to come out but not as much as the other yet; Could be a coincidence but maybe not.lets slow this runaway wagon down just a bit. when people talk about chasing numbers, this is a prime example of it. the situation that you are running in to is that you have two test methods that are giving you different results. one a manual test, and the otehr is an equipment test. your ph has reflected great on the manual test, and the probe test is not, and you have decided to disregard the manual test you did, for the sake of assuming that the probe test is the accurate of the two, and now youre buying scrubbers, buffers, etc to correct a number from a probe, that may not be showing right.
everythign from maybe buying plants and whatnot to fix pH that may not need to be fixed, if your manual testing shows a good pH. if thats the case, i have a bridge to sell you.
I have 100% faith the probe is now correct because I calibrated it and then stuck it into 7 and 10 solution and it read those numbers exactly at 7 and 10. I then ripped open a new packet and tested it again and it was spot on that time also.
So believing that my PH has been down around 7.4 for awhile (which explains a lot) and that even now it's constantly below 7.9 I think I have to act fast. A Calcium reactor is somwhere around 6.5 - 6.8 PH and that thing literally melts coral skeletons. I'm not trying to be anywhere near that number.
Also the Scrubber was only $37 from World Wide, Seachem Buffer $10 and the house plants I was going to get anyway.
More likely to be calibration issues than pH this low. This is what I do when I calibrate.
1. Check the expiration date on the packet. If it's expired up to 0.3 pH error is common.
2. Float both calibration packets in your sump for 15 minutes.
3. Perform a manual calibration
4. When doing the 7.0 step, rinse probe off in RODI water, pad dry with paper towel, insert into solution and shake/ tap any bubbles loose. Leave for 5 minutes and then click next.
5. Repeat step 4 but for the 10.0 solution.
6. Check probe in 7 and 10 solutions one at a time for 30 seconds they should be within 0.1pH. If they aren't, start over.
1. Check the expiration date on the packet. If it's expired up to 0.3 pH error is common.
2. Float both calibration packets in your sump for 15 minutes.
3. Perform a manual calibration
4. When doing the 7.0 step, rinse probe off in RODI water, pad dry with paper towel, insert into solution and shake/ tap any bubbles loose. Leave for 5 minutes and then click next.
5. Repeat step 4 but for the 10.0 solution.
6. Check probe in 7 and 10 solutions one at a time for 30 seconds they should be within 0.1pH. If they aren't, start over.
Someone suggested this last week (I did it then) and it worked perfectly. ThanksMore likely to be calibration issues than pH this low. This is what I do when I calibrate.
1. Check the expiration date on the packet. If it's expired up to 0.3 pH error is common.
2. Float both calibration packets in your sump for 15 minutes.
3. Perform a manual calibration
4. When doing the 7.0 step, rinse probe off in RODI water, pad dry with paper towel, insert into solution and shake/ tap any bubbles loose. Leave for 5 minutes and then click next.
5. Repeat step 4 but for the 10.0 solution.
6. Check probe in 7 and 10 solutions one at a time for 30 seconds they should be within 0.1pH. If they aren't, start over.
Better off with soda ash or sodium hydroxide. They have a larger pH increasing effect and are a lot cheaper.
right, but youre still discounting the fact that you have taken a manual test, got a reading that i, or most would not have a reason to doubt, doubted the outcome of that test for the sake of assuming that the lower result of the two is more accurate.I get what you're saying, but it's actually more to it. I have a pink/yellow and Red Gonipora that had been closed up for for weeks. What I didn't post was that once I got the CO2 Scrubber the very next day the pink one was fully extended. The Red one has started to come out but not as much as the other yet; Could be a coincidence but maybe not.
I have 100% faith the probe is now correct because I calibrated it and then stuck it into 7 and 10 solution and it read those numbers exactly at 7 and 10. I then ripped open a new packet and tested it again and it was spot on that time also.
So believing that my PH has been down around 7.4 for awhile (which explains a lot) and that even now it's constantly below 7.9 I think I have to act fast. A Calcium reactor is somwhere around 6.5 - 6.8 PH and that thing literally melts coral skeletons. I'm not trying to be anywhere near that number.
Also the Scrubber was only $37 from World Wide, Seachem Buffer $10 and the house plants I was going to get anyway.
if you do a test with the salifert kit 10 times, and come up with a proper reading, have someone else test it with their test kit, come up with the same reading, would you still disregard those results and believe the pH probe?
when i got my apex, the salnity didnt read right, but i didnt just disregard what a refractometer was telling me. the refractometer was telling me something different than what the pH probe was saying. if you had the salinity probe and it was doing what your pH probe is doing, would you do the same with its readings and disregard what the refractometer told you?
do what youre gonna do, but i dont know that i would trust the result of a new pH probe over a test kit outcome at this time, yet. especially if the manual test comes up with the same results multiple times.
if your pH probe is reading, lets say .3 too low, and the actual pH is 8.1, effectively you would be at about an 8.3-8.4 pH, so i wouldnt say its coincidence, but i would say that your coral like a higher pH. now, whether the pH probe is still right with its reading, is still unknown
Last edited:
Also every probe can be affected by "stray current". Basically every pump, heater and such in the tank will leak a small amount of current and also magnetic waves. This can affect your pH readings. Did you try to measure your DT water OUTSIDE the tank vs directly in the tank?
Yes, it was the same.Also every probe can be affected by "stray current". Basically every pump, heater and such in the tank will leak a small amount of current and also magnetic waves. This can affect your pH readings. Did you try to measure your DT water OUTSIDE the tank vs directly in the tank?
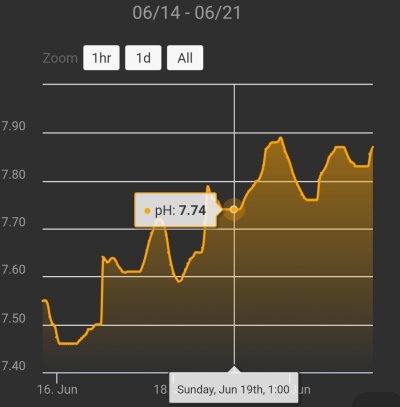
I mean just look at this test; It could easily be 7.89. Right?right, but youre still discounting the fact that you have taken a manual test, got a reading that i, or most would not have a reason to doubt, doubted the outcome of that test for the sake of assuming that the lower result of the two is more accurate.
if you do a test with the salifert kit 10 times, and come up with a proper reading, have someone else test it with their test kit, come up with the same reading, would you still disregard those results and believe the pH probe?
when i got my apex, the salnity didnt read right, but i didnt just disregard what a refractometer was telling me. the refractometer was telling me something different than what the pH probe was saying. if you had the salinity probe and it was doing what your pH probe is doing, would you do the same with its readings and disregard what the refractometer told you?
do what youre gonna do, but i dont know that i would trust the result of a new pH probe over a test kit outcome at this time, yet. especially if the manual test comes up with the same results multiple times.
if your pH probe is reading, lets say .3 too low, and the actual pH is 8.1, effectively you would be at about an 8.3-8.4 pH, so i wouldnt say its coincidence, but i would say that your coral like a higher pH. now, whether the pH probe is still right with its reading, is still unknown
Attachments
Just want to say everyone who has participated in this thread has been extremely helpful. Hopefully the info here will help someone else one day. I'll continue to update until I can consistently maintain a PH at or above 8.0.
it could be, but were also early in the day. my apex probe wont reach max pH until about 6pm. in fact, my pH right now is 7.97, so still, im not sure why youre chasing a number right now. on the further right two days, i had my windows open. this is to show the fact that the time in which you tested, is probably the reason for the lower number. if the probe matches the test, then i would suspect it has to do with the time of day. but, unless youre dosing something to keep it equal throughout the whole day, youre going to have high and lows. your highest wont be until at least the later part of the day. also to mention, the left two days i had a scrubber attached as well. the larger vertical lines is when i do my daily manual dose of alk. the furthest right day had a higher dose of alk than the day before. so thats why its kinda all over the place right now.I mean just look at this test; It could easily be 7.89. Right?
for example-
Last edited:
There must a reason why it goes to 7.8 pH with a scrubber and dK 12. It is either very litte gas exchange or wrong Alk/pH readings. I would not call this chasing numbers but problem-solving.it could be, but were also early in the day. my apex probe wont reach max pH until about 6pm. in fact, my pH right now is 7.97, so still, im not sure why youre chasing a number right now
Again, if its due to CO2 we are talking about harmful levels of indoor CO2 or no gas exchange in the tank. Otherwise one of the tests is performing poorly and deserves to be found out why
Do you see the media already consumed? It could last very little if you have a lot of indoor CO2.Yes it changes colors. You also have to take into consideration that I started around 7.4 so going up to 7.89 is a big jump (right?). Just not the jump I need. Not sure if running the AC affects PH, but mine is on all the time.
its guaranteed c02. thats what would cause low pH. but when i say chasing numbers, he is going off of a probe that is quite new, versus going off of a test kit that almost all people on this board would take as more accurate at this point. the issue is c02. guaranteed, but if were trying to correct a 7.8 pH early in the morning, who is to say that his pH isnt 8.15 during the peak time of the day since pH is guaranteed to fluctuate within 12 hours of the same day. were not even at what would be the highest pH time of the day. to combat that, youre gonna have to dose something during the low period, which were still in. but, if you want stability, i wouldnt go on dosing because your alk is already elevated as it is. if anything, i would go chaetoThere must a reason why it goes to 7.8 pH with a scrubber and dK 12. It is either very litte gas exchange or wrong Alk/pH readings. I would not call this chasing numbers but problem-solving.
Again, if its due to CO2 we are talking about harmful levels of indoor CO2 or no gas exchange in the tank. Otherwise one of the tests is performing poorly and deserves to be found out why
Last edited:
I would still try the aeration test with an airstone when temperatures are lower. And quantify how much pH difference you get outdoor vs indoor. You should get a pH of around 8.3 outside (500 ppm CO2) if this calculator is to be relied on https://www.hamzasreef.com/Contents/Calculators/EquilibriumPh.phpYou look earlier in the thread and I tested my indoor environment by opening the windows during the day times for two days. My PH level did increase, but it's 100+ degrees outside during Florida Summers so that's not an option. This is why I went and got the C02 Scrubber.
Also confirmed that the Probe is calibrated correctly. Earlier in this thread I got some great advice on how to calibrate the probe and its spot on with the calibration fluid now. Also bought some other PH Test kits just to confirm.
I've only been running the CO2 Scrubber for three days but I'm not sure this is something that improves with time. The PH numbers jump significantly with in the first few hours of running and have remained pretty constant since.
I know plants will probably have no affect but I was going to get some anyway. I'll be buying 5 Snake plants from home Depot Today.
I did see somewhere that when you top off none buffered RODI water to your aquarium that you are increasing the acidification of the tank. I tested my RODI yesterday after that and it's at a solid Zero TDS, hence my next comment.
I also ordered some Seachem Reef Buffer PH 8.3 last night from Amazon. Not sure if I'm going to use it but I want to have it on hand in case I'd do.
7.8 with lime soda. Is it me or I find this quite extreme?its guaranteed c02. thats what would cause low pH. but when i say chasing numbers, he is going off of a probe that is quite new, versus going off of a test kit that almost all people on this board would take as more accurate at this point. the issue is c02. guaranteed, but if were trying to correct a 7.8 pH early in the morning, who is to say that his pH isnt 8.15 during the peak time of the day since pH is guaranteed to fluctuate within 12 hours of the same day. were not even at what would be the highest pH time of the day.
There's people threads about struggling to keep pH LOW enough using CO2 scrubbers, with Alk levels much lower. I think there is something going on, at 2000ppm CO2 you start to feel "heavy" breathing, like when you are in winter in a packed car. That's unhealthy if spend a lot of time there
i would agree that 7.8 with lime is extreme, but it was also shown earlier that the probe didnt match the test kit, so if it isnt matching, which do we trust? the new probe, or a chemistry test that almost all people use to test their water. if we just assume that the probe is right, or close enough, is the level still low? of course, but dosing more to reach that, i think is beyond what should be considered. dosing the reef buffer is just raising kh more, just to get a pH number, and its already elevated higher than many people have theirs at already. so, i guess when i say chasing a number, im more into dosing with chemicals and adding things directly to the water of the tank to achieve a result instead of finding an underlying issue.7.8 with lime soda. Is it me or I find this quite extreme?
There's people threads about struggling to keep pH LOW enough using CO2 scrubbers, with Alk levels much lower. I think there is something going on, at 2000ppm CO2 you start to feel "heavy" breathing, like when you are in winter in a packed car. That's unhealthy if spend a lot of time there
Last edited:
Yes sorry I misinterpreted your comment. I totally agree, i never went through any comment on the forum about issues with pH related to the acidity of the RODI water. Also as @Randy Holmes-Farley explained on his website the main cause of low pH is to be found in CO2 levels, either inside or outside the tank.i would agree that 7.8 with lime is extreme, but it was also shown earlier that the probe didnt match the test kit, so if it isnt matching, which do we trust? the new probe, or a chemistry test that almost all people use to test their water. if we just assume that the probe is right, or close enough, is the level still low? of course, but dosing more to reach that, i think is beyond what should be considered. dosing the reef buffer is just raising kh more, just to get a pH number, and its already elevated higher than many people have theirs at already. so, i guess when i say chasing a number, im more into dosing with chemicals and adding things directly to the water of the tank to achieve a result instead of finding an underlying issue.
The salifert test he did was not looking green from the picture, but it's hard to tell from here. He should get a pH of 8.3 with normal CO2 levels+ scrubber + kh 12 so a dark green color on the test.
I don't know if someone else asked but I would try draw air for the skimmer from the outside and check before lights go on and before they start to ramp down. If you have the opportunity to seal the window and still run an airline outside, depending on the window structure/distance from the tank.
Similar threads
- Replies
- 2
- Views
- 178
- Replies
- 26
- Views
- 883
- Replies
- 18
- Views
- 547