Please, if you're using a solution of sodium carbonate, pH is not an issue! Testing will tell you what you'll need, but I'm thinking somewhere around 20 mls, if not more.....you have clams. Again testing will tell you what your daily consumption is.
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
New DIY Two Part Recipes with Higher pH Boost
- Thread starter Randy Holmes-Farley
- Start date
- Tagged users None
Just to be clear, you are mixing up just sodium carbonate, and not the "new" alk that includes sodium hydroxide.
Correct, I used sodium bicarbonate and baked it which leaves sodium carbonate. That's all I mixed for the alk part.
A.
Please, if you're using a solution of sodium carbonate, pH is not an issue! Testing will tell you what you'll need, but I'm thinking somewhere around 20 mls, if not more.....you have clams. Again testing will tell you what your daily consumption is.
Any ideas why my manual daily dose of baked sodium bicarbonate (sodium carbonate) isn't raising the alk in my tank?
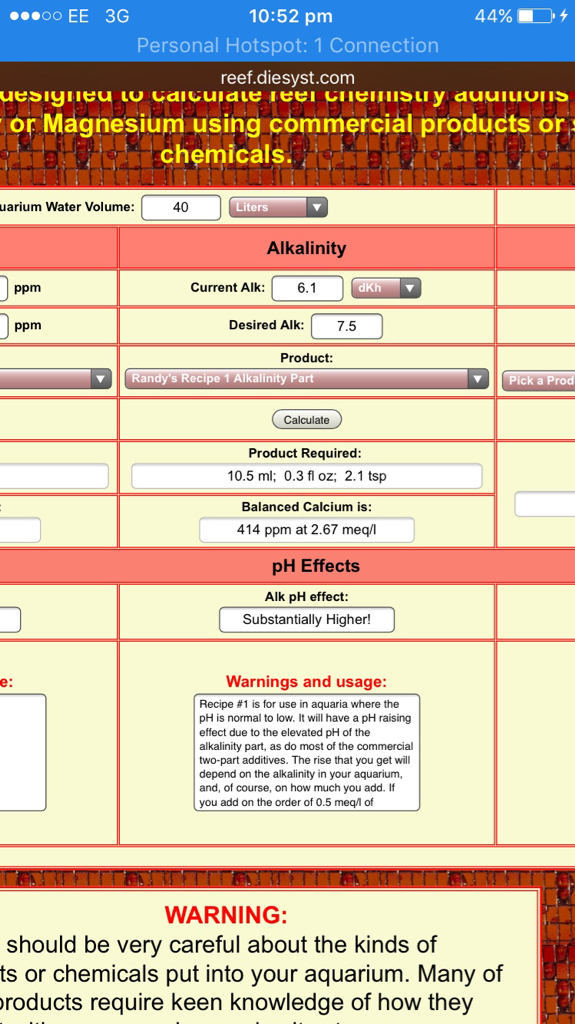
Tank volume after displacement 40L
Stock solution using baked bicarbonate (sodium carbonate) measured from 156g and then baked. Mixed into 1L rodi water. Using the dosing calc I'm led to believe to raise from current 6.1dKH to hit 7.5dKH I need 10.5ml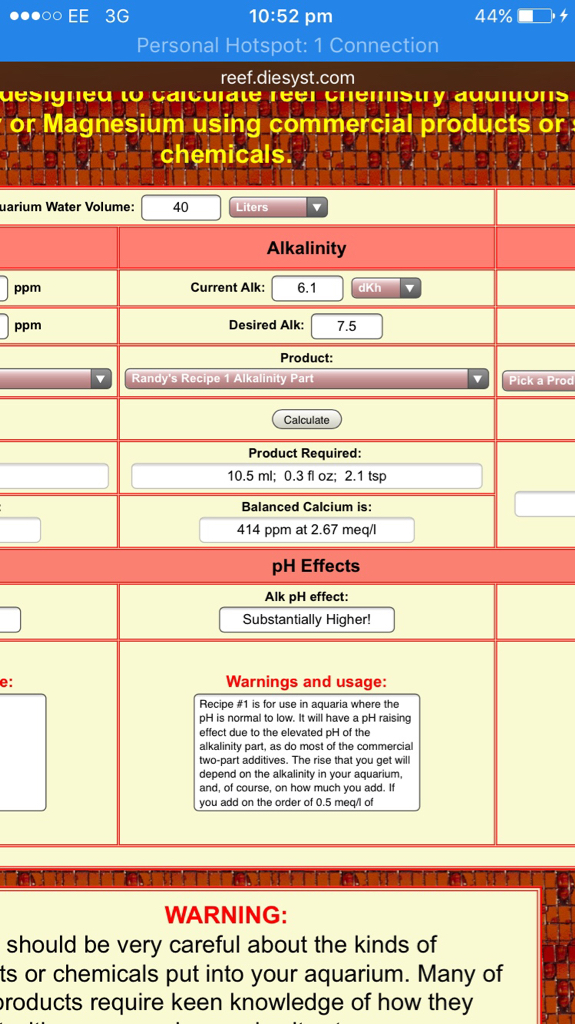
So for the last 2 days I've been Dosin 7ml/day (as to account for not raising ph too high too fast. However dKH has held at 6.1dKH.....
I'm not measuring ph as I've not got a ph tester, is it best i begin to dose unbaked bicarbonate as this doesn't raise the ph?
Thanks.
A.
I've filled the table like this,
Look correct?
A.
I've filled the table like this,
Look correct?
A.
I don't see anything.
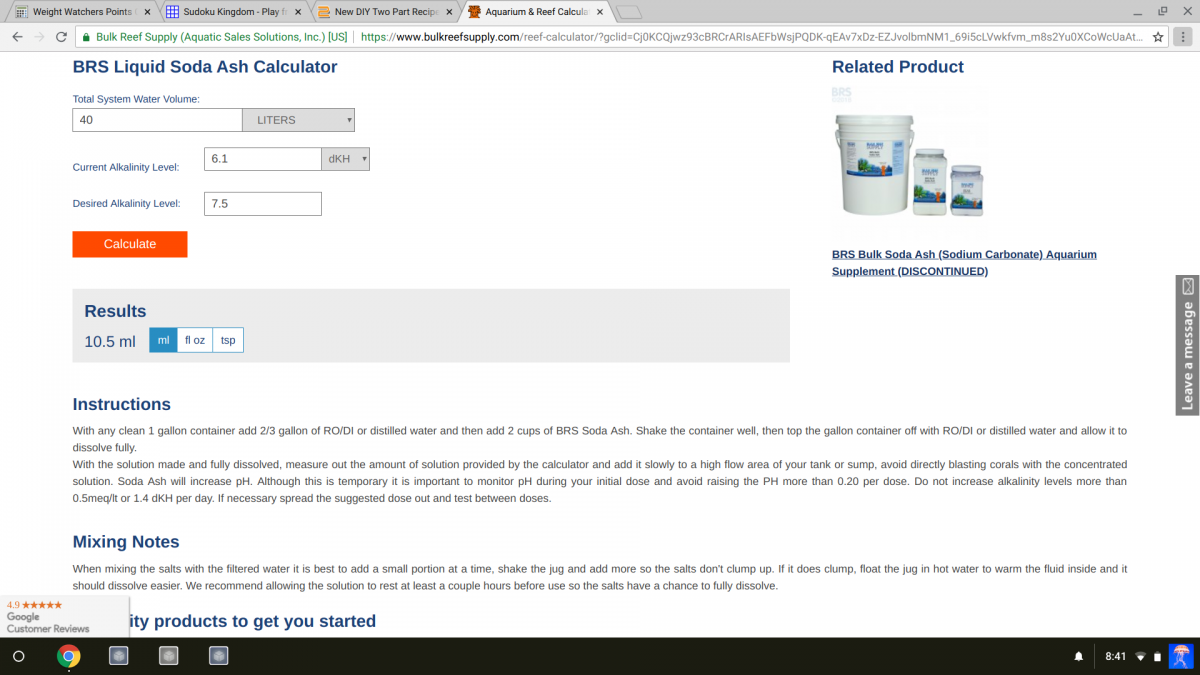
Looks spot on.....I verified with the BRS calculator.
But now you need to determine daily consumption, as I stated above. Might wish to look at THIS.
But now you need to determine daily consumption, as I stated above. Might wish to look at THIS.
- Joined
- Aug 15, 2016
- Messages
- 285
- Reaction score
- 90
Correct, I used sodium bicarbonate and baked it which leaves sodium carbonate. That's all I mixed for the alk part.
A.
If everything is baked 53gr sodiumcarbonate equals 84gr sodiumbicarbonate. Maybe not everything turned into sodiumcarbonate?
53gr dissolved in a liter of stock solution results in +2,8 kH per ml per liter aquariumwater (total solution = 2800kH per L aquariumwater). If you add 3ml of this stock solution to to 997ml of RODI it should result in 8.4 kH (if your test is accurate.. if you measured 53gr exactly... and if I am not mistaken).
Randy, or perhaps someone who might know. Would this work and be safe for dosing in my reef tank.

Randy, or perhaps someone who might know. Would this work and be safe for dosing in my reef tank.
It would probably be ok, but honestly we wouldn't know what impurities are in it unless someone tested the bag you get. 100% calcium chloride flake is no reflection on purity as we think of it in this application. I would personally go witb brs calcium chloride for a bit more money, or even you can get a 5 gallon bucket from drsfostersmith for cheaper. We have more options now for bulk calcium chloride at decent prices, I personally wouldn't use driveway heat or ice melt products in my tank.
You can get 35lbs from drsfostersmith right now for $60. That's pretty cheap. While we don't know for sure it's any more pure than the product in your picture, we can assume it's safe to use in our tanks.
chipmunkofdoom2
Always Making Something
View BadgesExcellence Award
Article Contributor
Reef Squad Emeritus
Wow, I'm led to believe carbonate raises ph substantially in comparison to bicarbonate!!! Going by comments and the calculator shown above in notes.
The pH change is not really that great with either supplement. Every 1.4 dKh increase with sodium carbonate raises the pH by about 0.3. Sodium bicarbonate, on the other hand, has a subtle pH lowering effect, but the effect is very small. For every 1.4 dKh increase with bicarb, the pH drops by 0.04. Depending on how well your pH probe is calibrated, you may not even be able to notice this decrease.
It's worth noting that if you keep your daily alkalinity increase to 0.5 - 1 dKh or lower, you'll only ever see a 0.1 - 0.2 pH increase with carbonate, or a 0.01 - 0.02 pH decrease with bicarb. Unless you're making really large changes, which aren't recommended anyway, the pH change is not a big concern.
- Joined
- Aug 15, 2016
- Messages
- 285
- Reaction score
- 90
Yesderday evening 19:00 I noticed my tank was clowdy. I quickly realised the tank was overdosed as the IKS doser continued to pump kH+ in my tank. The kH+ is a 2-mole NaOH strenght stock solution (80gr NaOH per liter stock). I believe 1,6 liter of stocksolution was dosed in 40min (dosing time should have been 1 min) to 400l tank water. The cause; defect timer.
The tank was clouded, brittle seastars and other animals trying to flee, corals covered in slime, tiny pest seastars where letting go of the glass.. a horrible sight. I dosed 200ml vinegar.. waited for a few minutes and tested kH, 12,5. I have muriatic acid.. dosed 40ml.. 30% strenght.. kH dropped to 9,3.. dosed another 10ml.. kH 8,6. Today many sps were brown.. measured kH which bounced back to 10.. however somehow they survived. I wonder if there is anything else I should do.. I have no way to measure pH.. it must have been astronomically high...
The tank was clouded, brittle seastars and other animals trying to flee, corals covered in slime, tiny pest seastars where letting go of the glass.. a horrible sight. I dosed 200ml vinegar.. waited for a few minutes and tested kH, 12,5. I have muriatic acid.. dosed 40ml.. 30% strenght.. kH dropped to 9,3.. dosed another 10ml.. kH 8,6. Today many sps were brown.. measured kH which bounced back to 10.. however somehow they survived. I wonder if there is anything else I should do.. I have no way to measure pH.. it must have been astronomically high...
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,529
- Reaction score
- 63,977
Sorry to hear this. keep us updated on how organisms fare.
The pH was likely in the 9's. There's a big buffering effect on the pH rise as magnesium hydroxide and calcium carbonate precipitate (luckily for us)
Unfortunately for us, the magnesium hydroxide that forms also buffers it as you try to lower it again, but the calcium carbonate does not since it won't redissolve.
I have no way to measure pH.. it must have been astronomically high...
The pH was likely in the 9's. There's a big buffering effect on the pH rise as magnesium hydroxide and calcium carbonate precipitate (luckily for us)
Unfortunately for us, the magnesium hydroxide that forms also buffers it as you try to lower it again, but the calcium carbonate does not since it won't redissolve.
- Joined
- Aug 15, 2016
- Messages
- 285
- Reaction score
- 90
A handfull of acropra browned. A few days before this happed, my black cucumber (which I have for 5 years) decided to crawl into the powerhead and release toxins which killed nearly all fish. There are not many organisms left.. The feeling of failure after failure... hurts. Spending all my time figuring out chemistry, diy projects etcetera everything went well.. and letting these relative simple things kill the tank within a week. I will keep you updated.. hopefully no one will make the same mistake...Sorry to hear this. keep us updated on how organisms fare.
The pH was likely in the 9's. There's a big buffering effect on the pH rise as magnesium hydroxide and calcium carbonate precipitate (luckily for us)
Unfortunately for us, the magnesium hydroxide that forms also buffers it as you try to lower it again, but the calcium carbonate does not since it won't redissolve.



Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,529
- Reaction score
- 63,977
Oh, so beautiful and so sad. 
If my pH is around 8.0-8.2, without addition of any kind...would I be able to use Recipe 1? I just don't want to commit to something, then find out it's not a match or screw something up.
Hi Randy, thanks a lot for your long term commitment to help us with reef aquaria chemistry.
I have a 120 g SPS tank with a long history of low pH. Typically pH goes down at night to 7,6-7.65 and goes up during the day to 7,9-7,95. I have the air intake of the skimmer connected to the outside, but even with that the pH very seldom goes beyond 8.0. For a long time I have been using a calcium reactor to keep alkalinity and calcium in place, helped by a kalwasser reactor which delivered kalk at nigh to help with pH.
When I heard about your new two-part with a pH boost I decided to give it a try. I didn't have sodium sulphate at hand so, just for the purpose of the trial, I prepared the alkalinity solution just with sodium hydroxide at 2X strength. The effect was terrific. For the last week my pH has ranged from 7,75-7,8 (night) to 8,15-8,2 (day) with a stable alkalinity (GHL KH Director). As soon as I got sodium sulphate I prepared a large batch of the number 2 solution, again at 2X strength. To my surprise, now the boosting effect on pH is mostly gone. My pH is now again more or less (may be a little higher, but just a little) in the old range. So, I have two questions:
1. Is it posible that the pH boosting effect is much higher with a sodium hydroxide solution alone than with the NaOH and NaSO4?
2. If the answer to this first question is positive, would it be too bad to remove the NaSO4 from the solution and leave the NaOH alone? I know there is a ionic imbalance involved here.
I have a 120 g SPS tank with a long history of low pH. Typically pH goes down at night to 7,6-7.65 and goes up during the day to 7,9-7,95. I have the air intake of the skimmer connected to the outside, but even with that the pH very seldom goes beyond 8.0. For a long time I have been using a calcium reactor to keep alkalinity and calcium in place, helped by a kalwasser reactor which delivered kalk at nigh to help with pH.
When I heard about your new two-part with a pH boost I decided to give it a try. I didn't have sodium sulphate at hand so, just for the purpose of the trial, I prepared the alkalinity solution just with sodium hydroxide at 2X strength. The effect was terrific. For the last week my pH has ranged from 7,75-7,8 (night) to 8,15-8,2 (day) with a stable alkalinity (GHL KH Director). As soon as I got sodium sulphate I prepared a large batch of the number 2 solution, again at 2X strength. To my surprise, now the boosting effect on pH is mostly gone. My pH is now again more or less (may be a little higher, but just a little) in the old range. So, I have two questions:
1. Is it posible that the pH boosting effect is much higher with a sodium hydroxide solution alone than with the NaOH and NaSO4?
2. If the answer to this first question is positive, would it be too bad to remove the NaSO4 from the solution and leave the NaOH alone? I know there is a ionic imbalance involved here.
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,529
- Reaction score
- 63,977
If my pH is around 8.0-8.2, without addition of any kind...would I be able to use Recipe 1? I just don't want to commit to something, then find out it's not a match or screw something up.
Yes, the pH is unlikely to get too high using a carbonate recipe.
Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,529
- Reaction score
- 63,977
Hi Randy, thanks a lot for your long term commitment to help us with reef aquaria chemistry.
I have a 120 g SPS tank with a long history of low pH. Typically pH goes down at night to 7,6-7.65 and goes up during the day to 7,9-7,95. I have the air intake of the skimmer connected to the outside, but even with that the pH very seldom goes beyond 8.0. For a long time I have been using a calcium reactor to keep alkalinity and calcium in place, helped by a kalwasser reactor which delivered kalk at nigh to help with pH.
When I heard about your new two-part with a pH boost I decided to give it a try. I didn't have sodium sulphate at hand so, just for the purpose of the trial, I prepared the alkalinity solution just with sodium hydroxide at 2X strength. The effect was terrific. For the last week my pH has ranged from 7,75-7,8 (night) to 8,15-8,2 (day) with a stable alkalinity (GHL KH Director). As soon as I got sodium sulphate I prepared a large batch of the number 2 solution, again at 2X strength. To my surprise, now the boosting effect on pH is mostly gone. My pH is now again more or less (may be a little higher, but just a little) in the old range. So, I have two questions:
1. Is it posible that the pH boosting effect is much higher with a sodium hydroxide solution alone than with the NaOH and NaSO4?
2. If the answer to this first question is positive, would it be too bad to remove the NaSO4 from the solution and leave the NaOH alone? I know there is a ionic imbalance involved here.
No to #1. Are you certain it was sodium sulfate and not sodium bisulfate?
It is ok to dose sodium hydroxide alone, which would be the same as dosing sodium carbonate or bicarbonate alone (except for pH).
Randy, is an aluminum cooking pan from the Dollar tree safe to cook my baking soda

Randy Holmes-Farley
Reef Chemist
View Badges
Staff member
Super Moderator
Excellence Award
Expert Contributor
Article Contributor
R2R Research
My Tank Thread
- Joined
- Sep 5, 2014
- Messages
- 67,529
- Reaction score
- 63,977
That should be ok, yes. Just don’t put any water into it.
Similar threads
- Replies
- 1
- Views
- 109
-
- AMS: Article
- Replies
- 61
- Views
- 4,363
- Replies
- 5
- Views
- 456
- Replies
- 8
- Views
- 158