In many threads – the nitrification process is discussed. This thread will try to handle how to construct the optimal nitrification filter for an aquarium. I´m aware that all does not have possibility to do an exactly copy of this, but the principles are general principles. It is important that this is a try to outline a filter that not exist in my aquarium – I have not try it – but I have tried to sum up my 50 years of experiences with this process. From aquariums, from wastewater treatments plants and from fish farms
Examination the nitrification process.
It is a 2-step process
Needs of the nitrification process
Both steps need surface to attach on
Both steps are very oxygen dependent. In fresh water there is studies showing that the first step can work at oxygen content of 3 mg/L O2, but the second step need at least 5 mg/L O2. Worth to mention here – saltwater content around 1.5 mg/L lesser O2 in the same temperature as freshwater – which means that saltwater at 25 degree C contain around 6.8 mg/L O2 at 100% saturation. Knowing of experiences that a reef aquarium with a good skimmer but no reversed refugium often run around 80 % saturation in nights (around 5.5 mg/L O2) is rather clear that the environment in our aquariums many times are a disadvantage for the nitrification cycle. If the second step stop or not work in the same rate as the first step – NO2 will be built up – something that often happens in newly started aquariums – often referred as “halted” nitrification. Please see this thread
Both steps (or more accurate the microorganism involved in the steps) need inorganic nutrients. N is no problem, but P can be a problem in newly started aquaria and has been reported as major cause for a stalling process – when adding PO4 – the second step started directly.
Both steps take inorganic carbon in form of CO3 (or HCO3) – they are alkalinity consumers. In freshwater – it can be a problem in water with low alkalinity (with following low pH – Low alkalinity water with pH below 7 stops the process – however is not the pH – it is the lack of CO3/HCO3 that stop the growth). In systems with adding CO2 for plant growth is normally recommended to use HCO3 in order to rise and retain the alkalinity around 2 in KH before starting the addition of CO2 in order to get a stable aquarium with working nitrification. In saltwater – no problems – the natural alkalinity is more than enough
For both steps - The organisms are slow growers compared to heterotrophic bacteria. Doubling time over 13 h compared with 15 to 30 minutes for heterotrophs
For both steps - After first establishment – they can go dormant for a while if the environment get hostile for them (an invasion of heterotrophs as an example). When the environment is good again, they come back rather fast in full strength
Summary – optimal conditions for nitrifiers
Advantages of this type of filter
Possible remedies for some of the disadvantages
Sincerely Lasse
Examination the nitrification process.
It is a 2-step process
- In the first step NH3/NH4 (ammonia gas or ammoniac/ammonia ion or ammonium) is oxidized (converted with help of oxygen) into nitrite by many types of autotrophic bacteria (AOB = Ammonia Oxidizing Bacteria) and archaea (AOA = Ammonia Oxidizing Archaea)
- In the second step NO2 (nitrite) is oxidized into NO3 (Nitrate) by a few autotrophic bacteria genera including Nitrobacter and Nitrospira (NOB = Nitrite Oxidizing Bacteria)
Needs of the nitrification process
Both steps need surface to attach on
Both steps are very oxygen dependent. In fresh water there is studies showing that the first step can work at oxygen content of 3 mg/L O2, but the second step need at least 5 mg/L O2. Worth to mention here – saltwater content around 1.5 mg/L lesser O2 in the same temperature as freshwater – which means that saltwater at 25 degree C contain around 6.8 mg/L O2 at 100% saturation. Knowing of experiences that a reef aquarium with a good skimmer but no reversed refugium often run around 80 % saturation in nights (around 5.5 mg/L O2) is rather clear that the environment in our aquariums many times are a disadvantage for the nitrification cycle. If the second step stop or not work in the same rate as the first step – NO2 will be built up – something that often happens in newly started aquariums – often referred as “halted” nitrification. Please see this thread
Both steps (or more accurate the microorganism involved in the steps) need inorganic nutrients. N is no problem, but P can be a problem in newly started aquaria and has been reported as major cause for a stalling process – when adding PO4 – the second step started directly.
Both steps take inorganic carbon in form of CO3 (or HCO3) – they are alkalinity consumers. In freshwater – it can be a problem in water with low alkalinity (with following low pH – Low alkalinity water with pH below 7 stops the process – however is not the pH – it is the lack of CO3/HCO3 that stop the growth). In systems with adding CO2 for plant growth is normally recommended to use HCO3 in order to rise and retain the alkalinity around 2 in KH before starting the addition of CO2 in order to get a stable aquarium with working nitrification. In saltwater – no problems – the natural alkalinity is more than enough
For both steps - The organisms are slow growers compared to heterotrophic bacteria. Doubling time over 13 h compared with 15 to 30 minutes for heterotrophs
For both steps - After first establishment – they can go dormant for a while if the environment get hostile for them (an invasion of heterotrophs as an example). When the environment is good again, they come back rather fast in full strength
Summary – optimal conditions for nitrifiers
- Enough space to colonize and maintain colonized and in direct contact with the streaming water
- No or few heterotrophs that compete about space
- Good oxygen concentration in the interface between biofilm and water
- NH3/NH4 (first step) and NO2 as energy resource
- Some PO4 in the water
- Alkalinity above 2 in KH
- A possibility for a fast flow that polishing the filter material
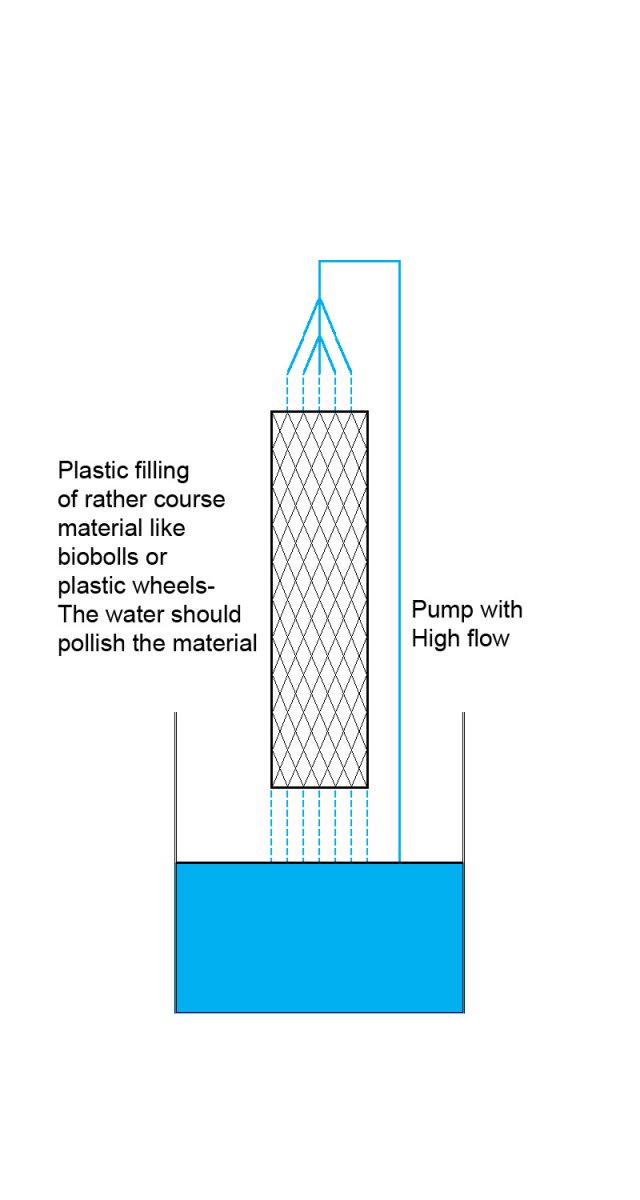
- The filter material should be course enough that it allows a thin interface between water and material all over the filter. Small pores should be avoided
- Free access for air in the whole filter
- Possibility to have a reversed flow of air through the filter – from the bottom up through the top
- Even spread of the water through the filter
- As high and maybe as narrow as possible allowing a strong stream through it
- No solid bottom - the water should not accumulate in the filter. Bottom of plastic grid as an example
- The water – flushed biofilms comes out into the water: If placed in sump – place it before the skimmer if you want the skimmer to take away the bacteria – if you want it up to your corals – place it before the return pump.
Advantages of this type of filter
- High rate of nitrification over time
- Because the filter material will not be in water if the pump stop – no risk of forming of hydrogen sulphide gas
- No cleaning or back flushes needed
- High gas exchange allowing ammonia gas (ammoniac) be aerated out. Oxygenate the water if the O2 level is below saturation and take out O2 if the water is supersaturated. CO2 – see below
- If the air has low content of CO2 (outside air or air scrubbed by a CO2 scrubber) it will rise the pH if it is low in the water (below 8.15 for outside air) and if the pH is above 8.15 (outside air) it will lower the pH (adding more CO2 to the system)
- If any there is any volatile gases in the room – it will transport them into the water up to the equilibrium point for this gas and water.
- If the room have high content of CO2 – it will lower the pH – ventilation with outside air may help
- Need space and height
- May remove all NH3/NH4 too quick and it could harm some organisms that use photosynthesis (the ones that can´t handle NO3 as a source for inorganic nitrogen)
- It will be a faster evaporation of water and require more top off (Thank you @Miller535 for that input)
- the tower will act as an heat exchanger - it can be both a advantage/disadvantage depended of temperature difference between the aquarium and the air used in the tower.
Possible remedies for some of the disadvantages
- Because the filter material will not be in water if the pump stop – it is possible to let the pH manage the filter and also only use it during 2-4 hours after feeding when the load of free NH3/NH4 is largest
Sincerely Lasse
Last edited:

















