Merry Christmas to all the staff and leaders at the best public aquarium on R2R! My your new year be excellent!
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Swedish fish - behind the scenes rebuilding a public aquarium
- Thread starter Sallstrom
- Start date
- Tagged users None
- Status
- Not open for further replies.
crusso1993
7500 Club Member
View BadgesTampa Bay Reef Keepers
West Palm Beach Reefer
Hospitality Award
Ocala Reef Club Member
MAC of SW Florida
Wishing good tidings to you and the whole school at the aquarium!

Fudsey
Jack of all trades, Master of none ;-)
View Badges
Reef Squad
Excellence Award
Reef Tank 365
Photo of the Month
Hospitality Award
NHFS Member
My Tank Thread
Merry Christmas Sallstrom and all at the aquarium ! !
merry christmas to everyone!
I know many of you like pumps  so here’s a short post on how we restored/upgraded a Deltec skimmer (SC 3070 internal I think is the name of the skimmer).
so here’s a short post on how we restored/upgraded a Deltec skimmer (SC 3070 internal I think is the name of the skimmer).
First I ordered new DC pumps directly from Deltec, and asked for pumps that would fit in their skimmer. The old pumps were AC and we want to switch over to DC pumps.
It turned out that we still needed to rebuild the outlet(after the pumps, in the skimmer), but it just took some pvc glue and 5 minutes of work.
The pumps look very much like a another well known brand...
The air tubes had to be extended a bit too.


Next we needed to fix a new way to control the water level inside the skimmer, because we broke the old screw thing that was on the skimmer outlet.
I tried a very inexpensive method, and it worked out fine

A pvc pipe with a 45 degree ending to put inside the outlet pipe.

The outlet “fully opened “

The outlet closed(but not 100%). Just turned the pvc pipe 180 degrees.
First I ordered new DC pumps directly from Deltec, and asked for pumps that would fit in their skimmer. The old pumps were AC and we want to switch over to DC pumps.
It turned out that we still needed to rebuild the outlet(after the pumps, in the skimmer), but it just took some pvc glue and 5 minutes of work.
The pumps look very much like a another well known brand...
The air tubes had to be extended a bit too.
Next we needed to fix a new way to control the water level inside the skimmer, because we broke the old screw thing that was on the skimmer outlet.
I tried a very inexpensive method, and it worked out fine
A pvc pipe with a 45 degree ending to put inside the outlet pipe.
The outlet “fully opened “
The outlet closed(but not 100%). Just turned the pvc pipe 180 degrees.
- Joined
- May 19, 2017
- Messages
- 631
- Reaction score
- 434
Running both Triton Core7 and calcium reactors on both systems, to balance CO2 and pH.
What is the trick to finding the right balance when you add carx to the mix with Triton's method? Do you just handle X% of your demand with Trition and the rest with the calcium reactor? Or do you base it around a particular trace element and judge correct dosage around depletion rate from several ICP tests? Can't quite wrap my head around equilibrium
What is the trick to finding the right balance when you add carx to the mix with Triton's method? Do you just handle X% of your demand with Trition and the rest with the calcium reactor? Or do you base it around a particular trace element and judge correct dosage around depletion rate from several ICP tests? Can't quite wrap my head around equilibrium
In these temporary tanks we started with only Core7. When we closed our old tanks we also installed calcium rectors. What we did in this case was to just start them up, lower Core7, and meassure KH a lot. As a security we have a GHL KH Director which we could switch between the two coral systems just to give us an idea if the doses was too high or low.
After a while we sat Core7 to a fixed number(about 40 or 50% of the original dose when we only used Core7), and only change the reactors if we needed to get the KH up. So yes, Core7 takes care of about 50% of the demend at the moment.
We've used Calcium reactors and Triton ICP tests for five years or more, so we know that the reactors do a good job adding most of the elements. Core7 does the same job, but without the CO2.
Now we do the same as before, we fix the numbers that need adjusting with Triton supplements after getting the results from the ICP tests. But what I've seen from the first months with these new systems, we don't need to add a lot.
There are a couple of reasons why we run the tanks this way. One is to add CO2 and get a pH somewhere "in the middle", instead of very high or low. Another one is to lower the consumption of Core7.
Let me know if I there's anything more you would like to know!
- Joined
- May 19, 2017
- Messages
- 631
- Reaction score
- 434
Let me know if I there's anything more you would like to know!
Haha I could pick your brain for days.
We've used Calcium reactors and Triton ICP tests for five years or more, so we know that the reactors do a good job adding most of the elements. Core7 does the same job, but without the CO2.
Now we do the same as before, we fix the numbers that need adjusting with Triton supplements after getting the results from the ICP tests. But what I've seen from the first months with these new systems, we don't need to add a lot.
There are a couple of reasons why we run the tanks this way. One is to add CO2 and get a pH somewhere "in the middle", instead of very high or low. Another one is to lower the consumption of Core7.
Invaluable information =) Thanks for sharing. It seems like the best of both worlds - awesome to know it is!
Brew12
Electrical Gru
View BadgesExcellence Award
Reef Tank 365
Article Contributor
Moderator Emeritus
North Alabama Reef Club
Article Administrator
My Tank Thread
I've always thought calcium reactors add most trace elements but I've had some people tell me they add very little and that most trace elements are in the coral flesh and polyps. So you do feel that many of them are captured in the coral skeletons?We've used Calcium reactors and Triton ICP tests for five years or more, so we know that the reactors do a good job adding most of the elements.
Sorry, what I meant was macro elements. So from what I've seen Ca, Mg, B, Sr, K, and perhaps some more macro elemets are added from a calcium reactor(like PI've always thought calcium reactors add most trace elements but I've had some people tell me they add very little and that most trace elements are in the coral flesh and polyps. So you do feel that many of them are captured in the coral skeletons?
And running with only Core7 we had to add a lot of Sr.
So I think you need to do ICP tests(at Triton of course
And I guess you could get different results with different types of calcium reactor media. We use mostly large coral gravel.
Trace elements we dose seperately. Sorry for a long answer..
Brew12
Electrical Gru
View BadgesExcellence Award
Reef Tank 365
Article Contributor
Moderator Emeritus
North Alabama Reef Club
Article Administrator
My Tank Thread
I would have considered B, Sr, K and I to be trace elements. Not sure if that is scientifically correct or if there is even a clear delineation between what is a trace and what is a macro element.Sorry, what I meant was macro elements. So from what I've seen Ca, Mg, B, Sr, K, and perhaps some more macro elemets are added from a calcium reactor(like P). Not always in a perfect match to the demand from the tank in question. In the long run we had to add CaCl now and then for example, and a lot of B.
And running with only Core7 we had to add a lot of Sr.
So I think you need to do ICP tests(at Triton of course) regulary to figure out how the demand is in your tank. I adjusted the Core7 2 dosing yesterday because Ca started to raise in one of our coral systems. We have a large part of that system for soft corals, so the demand isn't the same as the other more stony coral dominated.
And I guess you could get different results with different types of calcium reactor media. We use mostly large coral gravel.
Trace elements we dose seperately. Sorry for a long answer..
I keep meaning to bring a chunk of dead coral in to the lab at work and running it under an electron scanning microscope. That way I can definitively see what exactly gets trapped in a coral skeleton.
Here are a quote from a Stanford lecture I just found:I would have considered B, Sr, K and I to be trace elements. Not sure if that is scientifically correct or if there is even a clear delineation between what is a trace and what is a macro element.
I keep meaning to bring a chunk of dead coral in to the lab at work and running it under an electron scanning microscope. That way I can definitively see what exactly gets trapped in a coral skeleton.
"Trace Elements – Those elements that do not contribute to the salinity. All elements are present in concentrations less than 1mg kg-1"
Yes, do that! That would be really cool to see. I know there are some old papers on that, but do it anyway!
Brew12
Electrical Gru
View BadgesExcellence Award
Reef Tank 365
Article Contributor
Moderator Emeritus
North Alabama Reef Club
Article Administrator
My Tank Thread
So the definition of trace elements in sea water would also apply when used to describe trace elements in coral?Here are a quote from a Stanford lecture I just found:
"Trace Elements – Those elements that do not contribute to the salinity. All elements are present in concentrations less than 1mg kg-1"
Yes, do that! That would be really cool to see. I know there are some old papers on that, but do it anyway!
Sorry for the late answer. I don’t knowSo the definition of trace elements in sea water would also apply when used to describe trace elements in coral?
For example.Haha I could pick your brain for days.
Invaluable information =) Thanks for sharing. It seems like the best of both worlds - awesome to know it is!
Here’s our Triton additives for corrections for January. The coral systems are about 4000L each.
On this list is not Mn and Fe which we add daily.
- Joined
- Jun 15, 2015
- Messages
- 257
- Reaction score
- 235
Hi.For example.
Here’s our Triton additives for corrections for January. The coral systems are about 4000L each.
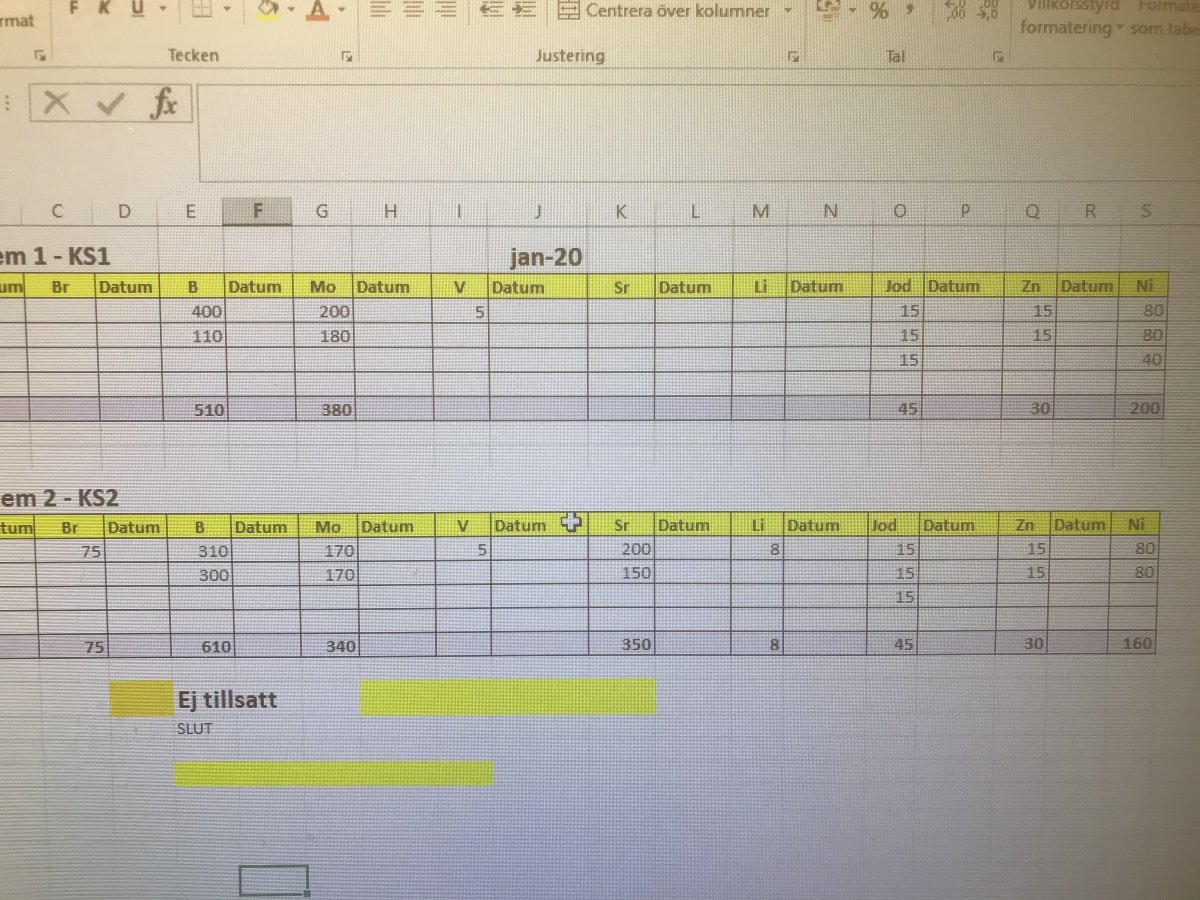
On this list is not Mn and Fe which we add daily.
May I ask which Mn and Fe aditives do you use? DIY?
And at what concentrations you daily dose each in ppb?
Thanks.
crusso1993
7500 Club Member
View BadgesTampa Bay Reef Keepers
West Palm Beach Reefer
Hospitality Award
Ocala Reef Club Member
MAC of SW Florida
Hi.
May I ask which Mn and Fe aditives do you use? DIY?
And at what concentrations you daily dose each in ppb?
Thanks.
Knowing that @Sallstrom is a crazy Swede and also knowing he hangs out with crazier Swede @Lasse , I'm inclined to think he just throws a few chunks of this into his sumps!
Would a spectrometer help, also? With coral skeleton. ....I would have considered B, Sr, K and I to be trace elements. Not sure if that is scientifically correct or if there is even a clear delineation between what is a trace and what is a macro element.
I keep meaning to bring a chunk of dead coral in to the lab at work and running it under an electron scanning microscope. That way I can definitively see what exactly gets trapped in a coral skeleton.
Looks like chunk of iron. Or fossilized dino doo doo.....Knowing that @Sallstrom is a crazy Swede and also knowing he hangs out with crazier Swede @Lasse , I'm inclined to think he just throws a few chunks of this into his sumps!

We use Triton's addetives for Fe and Mn, so I don't know the concentration. We follow Tritons instructions for dosage, but have adjusted them a bit after years of use.Hi.
May I ask which Mn and Fe aditives do you use? DIY?
And at what concentrations you daily dose each in ppb?
Thanks.
At the moments we dose Mn 8 ml/day and Fe 2ml/day, in each coral system.
- Status
- Not open for further replies.
Similar threads
-
- Poll
- Replies
- 18
- Views
- 1,141
- Replies
- 22
- Views
- 1,563
-
- Poll
- Replies
- 54
- Views
- 2,071
- Replies
- 29
- Views
- 1,027
- Replies
- 2
- Views
- 284














