Would it not be possible to grind some of the base away to make it fit in the chamber?I wanted to try one, So I grabbed the D size for my 25 gallon based on the information on the website. Theshrimptank.
I wish I would have looked for sizing on each unit as the D size fits my water volume, but not in the middle chamber of my AIO like I was hoping. I will fit in the Display, so I'll probably keep it in case of an emergency power outage or something, plus I emailed the company and never got a response to exchange it for the "nano" version.
Everything I've seen is very positive, I should probably just order the nano and give it a try, just not sure on its overall size.
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Anyone using Dr. Sochting's Oxydator
- Thread starter PapaDragon
- Start date
- Tagged users None
As if you don't have much to do - do you think it might be helpful to write up a little protocol of what / how to use H2O2 during a power outage?:). Please?To add H2O2 to indoor fish farm tanks during a power breakdown have saved many tons of fish for me. Its not snake oil at all
Sincerely Lasse
PS - on the oxydator - mechanism of action
1. With the Orange side - with the 2 holes down - which happens:?
a. The catalyst causes O2 to push up into the top - and the H2O2 comes out the bottom
b. Another mechanism.
I was not under the complete impression that the Oxydator released Oxygen into the tank - but rather - H2O2 - which is rapidly turned into H20 and O2 EDIT - pushes H2O2 out of the reservoir
1. With the Orange side - with the 2 holes down - which happens:?
a. The catalyst causes O2 to push up into the top - and the H2O2 comes out the bottom
b. Another mechanism.
I was not under the complete impression that the Oxydator released Oxygen into the tank - but rather - H2O2 - which is rapidly turned into H20 and O2 EDIT - pushes H2O2 out of the reservoir
Last edited:
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
Yest it release H2O2 out and it will come i direct contact with the secondary catalyst - the ceramic around the plastic bottle. Here - pure O2 will be produced. If the concentration and/or the catalysts inside the bottle is to many. The outcoming H2O2 have not enough contact time with the second catalyst and pure h2O2 will come out in the aquarium. This can be good when you try to defend a parasite or a disease.was not under the complete impression that the Oxydator released Oxygen into the tank - but rather - H2O2 - which is rapidly turned into H20 and O2
sincerely Lasse
PS - on the oxydator - mechanism of action
1. With the Orange side - with the 2 holes down - which happens:?
a. The catalyst causes O2 to push up into the top - and the H2O2 comes out the bottom
b. Another mechanism.
I was not under the complete impression that the Oxydator released Oxygen into the tank - but rather - H2O2 - which is rapidly turned into H20 and O2
You might find the screen shot of a reply from Uwe of Sochtting on the very subject that was sent onto me by the UK distributor to clear up any confusion on how an Oxydator works.
Thansk Lasse:). I Meant - once the H2O2 contacts the catalyst - it will then cause O2 be released - which will go upwards into the container - forcing H2O2 out. It seems like mine runs out quickly:)Yest it release H2O2 out and it will come i direct contact with the secondary catalyst - the ceramic around the plastic bottle. Here - pure O2 will be produced. If the concentration and/or the catalysts inside the bottle is to many. The outcoming H2O2 have not enough contact time with the second catalyst and pure h2O2 will come out in the aquarium. This can be good when you try to defend a parasite or a disease.
sincerely Lasse
I figured it out about 30 years ago ;>)Thanks this is the only way I could figure it would work.
I thought I had as well :) just wanted to make sure - I am concerned how quickly my H2O2 is depletedI figured it out about 30 years ago ;>)
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
Go down in concentration or number of catalyst.
Sincerely Lasse
Sincerely Lasse
I did not know about the secondary catalyst!!!!!!!!!! should that be kept quite clean then??Yest it release H2O2 out and it will come i direct contact with the secondary catalyst - the ceramic around the plastic bottle. Here - pure O2 will be produced. If the concentration and/or the catalysts inside the bottle is to many. The outcoming H2O2 have not enough contact time with the second catalyst and pure h2O2 will come out in the aquarium. This can be good when you try to defend a parasite or a disease.
sincerely Lasse
Lasse
10K Club member
View Badges
Ultimate Member
Reef Squad
Excellence Award
Reef Tank 365
Reef Of The Month
R2R App User
Article Contributor
My Tank Thread
I did not know about the secondary catalyst!!!!!!!!!! should that be kept quite clean then??
Sincerely Lasse
Installed a model D in my ~25G total volume system, 6% with one catalyst. Some bubbles but not enough to worry...


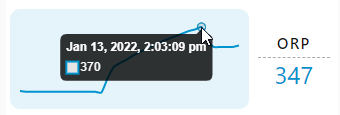
Can you explain - when you put it to work? Was is it was at 289 - ? Not sure what the graphs mean:). but thanks for postingInstalled a model D in my ~25G total volume system, 6% with one catalyst. Some bubbles but not enough to worry...



Can you explain - when you put it to work? Was is it was at 289 - ? Not sure what the graphs mean:). but thanks for posting
Graph shows the spike in ORP when initially adding it to my sump. ORP has been hovering around 280 during the day.
I was wondering why there are 3 graphs with different spikes? In other words what are the 3 graphs you posted?Graph shows the spike in ORP when initially adding it to my sump. ORP has been hovering around 280 during the day.
@CDNReefer got it. Wanted to show the time stamp for those curious. These aren’t important just showing an effect of adding the oxydator.I was wondering why there are 3 graphs with different spikes? In other words what are the 3 graphs you posted?
Sorry you lost me. all good - good luck with your oxydatorSame graphs , just look at the points not the number on the right
So you posted 3 different graphs - showing the same thing - (not sure how you did it - unless you had 3 different monitoring devices). I still don't get the POINT you're trying to make - so - the Orp went up - then went down.
He went from 289 to 370 with the addition of the oxydator. That's what I'm seeing. The point is it went up.
You all got me thinking again about the gallon of 12% I've got in my fridge. It's been in there for 6 months and I still have half left. How do you all generally determine if your peroxide is losing strength? A trending orp probe? The time in between refills? I see there are test strips available on Amazon.
You all got me thinking again about the gallon of 12% I've got in my fridge. It's been in there for 6 months and I still have half left. How do you all generally determine if your peroxide is losing strength? A trending orp probe? The time in between refills? I see there are test strips available on Amazon.
Similar threads
- Replies
- 47
- Views
- 1,784
- Replies
- 20
- Views
- 702
TOP 10 Trending Threads
-
- Sticky
- Replies
- 92
- Views
- 884
- Replies
- 61
- Views
- 1,380
- Replies
- 27
- Views
- 476
- Replies
- 32
- Views
- 638
- Replies
- 40
- Views
- 1,350
- Replies
- 57
- Views
- 693
- Replies
- 61
- Views
- 1,277
- Replies
- 22
- Views
- 396
New Posts
-
Cycling Help - What is the best options for me
- Latest: Ryan - Serious Reefs

















